Superalloy
A superalloy, or high-performance alloy, is an alloy with the ability to operate at a high fraction of its melting point.[1] Several key characteristics of a superalloy are excellent mechanical strength, resistance to thermal creep deformation, good surface stability, and resistance to corrosion or oxidation.

The crystal structure is typically face-centered cubic (FCC) austenitic. Examples of such alloys are Hastelloy, Inconel, Waspaloy, Rene alloys, Incoloy, MP98T, TMS alloys, and CMSX single crystal alloys.
Superalloy development has relied heavily on both chemical and process innovations. Superalloys develop high temperature strength through solid solution strengthening and precipitation strengthening from secondary phase precipitates such as gamma prime and carbides. Oxidation or corrosion resistance is provided by elements such as aluminium and chromium. Superalloys are often cast as a single crystal—while grain boundaries may provide strength at low temperatures, they decrease creep resistance.
The primary application for such alloys is in aerospace and marine turbine engines. Creep is typically the lifetime-limiting factor in gas turbine blades.[2]
Superalloys are the materials which have made much of very-high-temperature engineering technology possible.[3]
Chemical development
Because these alloys are intended for high temperature applications (i.e. holding their shape at temperatures near their melting point) their creep and oxidation resistance are of primary importance. Nickel (Ni) based superalloys have emerged as the material of choice for these applications because of their unique γ' precipitates.[1][4] The properties of these Ni based superalloys can be tailored to a certain extent through the addition of many other elements, both common and exotic, including not only metals, but also metalloids and nonmetals; chromium, iron, cobalt, molybdenum, tungsten, tantalum, aluminium, titanium, zirconium, niobium, rhenium, yttrium, vanadium, carbon, boron or hafnium are some examples of the alloying additions used. Each of these additions has been chosen to serve a particular purpose in optimizing the properties for high temperature application.
Creep resistance is dependent, in part, on slowing the speed of dislocation motion within a crystal structure. In modern Ni based superalloys, the γ’-Ni3(Al,Ti) phase present acts as a barrier to dislocation motion. For this reason, this γ’ intermetallic phase, when present in high volume fractions, drastically increases the strength of these alloys due to its ordered nature and high coherency with the γ matrix. The chemical additions of aluminum and titanium promote the creation of the γ’ phase. The γ’ phase size can be precisely controlled by careful precipitation strengthening heat treatments. Many superalloys are produced using a two-phase heat treatment that creates a dispersion of cuboidal γ’ particles known as the primary phase, with a fine dispersion between these known as secondary γ’. In order to improve the oxidation resistance of these alloys, Al, Cr, B, and Y are added. The Al and Cr form oxide layers that passivate the surface and protect the superalloy from further oxidation while B and Y are used to improve the adhesion of this oxide scale to the substrate.[5] Cr, Fe, Co, Mo and Re all preferentially partition to the γ matrix while Al, Ti, Nb, Ta, and V preferentially partition to the γ’ precipitates and solid solution strengthen the matrix and precipitates respectively. In addition to solid solution strengthening, if grain boundaries are present, certain elements are chosen for grain boundary strengthening. B and Zr tend to segregate to the grain boundaries which reduces the grain boundary energy and results in better grain boundary cohesion and ductility.[6] Another form of grain boundary strengthening is achieved through the addition of C and a carbide former, such as Cr, Mo, W, Nb, Ta, Ti, or Hf, which drives precipitation of carbides at grain boundaries and thereby reduces grain boundary sliding.
| Element | Composition range (weight %) |
Purpose |
|---|---|---|
| Ni, Fe, Co | 50-70% | These elements form the base matrix γ phase of the superalloy. Ni is necessary because it also forms γ' (Ni3Al). Fe and Co have higher melting points than Ni and offer solid solution strengthening. Fe is also much cheaper than Ni or Co. |
| Cr | 5-20% | Cr is necessary for oxidation and corrosion resistance; it forms a protective oxide Cr2O3 |
| Al | 0.5-6% | Al is the main γ' former. It also forms a protective oxide Al2O3, which provides oxidation resistance at higher temperature than Cr2O3 |
| Ti | 1-4% | Ti forms γ' |
| C | 0.05-0.2% | MC and M23C6 (M = metal) carbides are the strengthening phase in the absence of γ' |
| B,Zr | 0-0.1% | Boron and zirconium provide strength to grain boundaries. This is not essential in single-crystal tubine blades, because there are no grain boundaries |
| Nb | 0-5% | Nb can form γ'', a strengthening phase at lower (below 700 °C) temperatures |
| Re, W, Hf, Mo, Ta | 1-10% | Refractory metals, added in small amounts for solid solution strengthening (and carbide formation). They are heavy, but have extremely high melting points |
Active research
While Ni-based superalloys are excellent high temperature materials and have proven very useful, Co-based superalloys potentially possess superior hot corrosion, oxidation, and wear resistance as compared to Ni-based superalloys. For this reason, efforts have also been put into developing Co-based superalloys over the past several years. Despite that, traditional Co-based superalloys have not found widespread usage because they have a lower strength at high temperature than Ni-based superalloys.[9] The main reason for this is that—until recently—they appeared to lack the γ’ precipitation strengthening that is so important in the high temperature strength of Ni-based superalloys. A 2006 report on metastable γ’-Co3(Al,W) intermetallic compound with the L12 structure suggests Co based alloys as alternative to traditional Ni based superalloys. However this class of alloys was reported in a PhD thesis by C. S. Lee in 1971.[10] The two-phase microstructure consists of cuboidal γ’ precipitates embedded in a continuous γ matrix and is therefore morphologically identical to the microstructure observed in Ni based superalloys. Like in the Ni-based system, there is a high degree of coherency between the two phases which is one of the main factors resulting in the superior strength at high temperatures.
This provides a pathway for the development of a new class of load-bearing Co-based superalloys for application in severe environments.[11] In these alloys, W is the crucial addition for forming the γ’ intermetallic compound; this makes them much denser (>9.6 g/cm3) compared to Ni-based superalloys. Recently a new class of γ - γ’ cobalt based superalloys have been developed that are W-free and have much lower density comparable to nickel based superalloys.[12][13][14][15] In addition to the fact that many of the properties of these new Co-based superalloys could be better than those of the more traditional Ni-based ones, Co also has a higher melting temperature than Ni. Therefore, if the high temperature strength could be improved, the development of novel Co based superalloys could allow for an increase in jet engine operation temperature resulting in an increased efficiency.
Phase formation
Adding new elements is usually good because of solid solution strengthening, but engineers need to be careful about which phases precipitate. Precipitates can be classified as geometrically close-packed (GCP), topologically close-packed (TCP), or carbides. GCP phases are usually good for mechanical properties, but TCP phases are often deleterious. Because TCP phases are not truly close packed, they have few slip systems and are very brittle. They are additionally bad because they "scavenge" elements away from GCP phases. Many elements that are good for forming γ' or have great solid solution strengthening may precipitate TCPs. Engineers need to find the balance that promotes GCPs while avoiding TCPs.
An area of the alloy with TCP phase formation will be weak because[16][17]
- the TCP phase has inherently poor mechanical properties
- the TCP phase is incoherent with the γ matrix
- the TCP phase is surrounded by a "depletion zone" where there is no γ'
- the TCP phase usually forms sharp plate or needle-like morphologies which easily nucleate cracks
The main GCP phase is γ'. Almost all superalloys are Ni-based because of this phase. γ' is an ordered L12 (pronounced L-one-two), which means it has a certain atom on the face of the unit cell, and a certain atom on the corners of the unit cell. For Ni-based superalloys, that usually means Ni on the faces and Ti or Al on the corners.
Another "good" GCP phase is γ''. It is also coherent with γ, but it dissolves at high temperatures.
| Phase | Classification | Structure | Composition(s) | Appearance | Effect |
|---|---|---|---|---|---|
| γ | matrix | disordered FCC | Ni, Co, Fe and other elements in solid solution | The background for other precipitates | The matrix phase, provides ductility and a structure for precipitates |
| γ' | GCP | L12 (ordered FCC) | Ni3(Al,Ti) | cubes, rounded cubes, spheres, or platelets (depending on lattice mismatch) | The main strengthening phase. γ' is coherent with γ, which allows for ductility. |
| Carbide | Carbide | FCC | mC, m23C6, and m6C (m = metal) | string-like clumps, like strings of pearls | There are many carbides, but they all provide dispersion strengthening and grain boundary stabilization |
| γ'' | GCP | D022 (ordered BCT) | Ni3Nb | very small disks | This precipitate is coherent with γ'. It is the main strengthening phase in IN-718, but γ'' dissolves at high temperatures |
| η | GCP | D024 (ordered HCP) | Ni3Ti | may form cellular or Widmanstätten patterns | The phase is not the worst, but it's not as good as γ'. It an be useful in controlling grain boundaries |
| δ | not close-packed | orthorhombic | Ni3Nb | acicular (needle-like) | The main issue with this phase is that it's not coherent with γ, but it is not inherently weak. It typically forms from decomposing γ'', but sometimes it's intentionally added in small amounts for grain boundary refinement |
| σ | TCP | tetrahedral | FeCr, FeCrMo, CrCo | elongaged globules | This TCP is usually considered to have the worst mechanical properties.[18] It is never desirable for mechanical properties |
| μ | TCP | hexagonal | Fe2Nb, Co2Ti, Fe2Ti | globules or platelets | This phase has typical TCP issues. It is never desirable for mechanical properties |
| Laves | TCP | rhombohedral | (Fe,Co)7(Mo,W)6 | coarse Widmanstätten platelets | This phase has typical TCP issues. It is never desirable for mechanical properties |
Families of superalloys
History and development of Ni-based superalloys
The United States became interested in gas turbine development around 1905.[1] From 1910-1915, austenitic ( γ phase) stainless steels were developed for the high temperatures in gas turbines. By 1929, 80Ni-20Cr alloy was the norm, with small additions of Ti an Al. Although early metallurgists did not know it yet, they were forming small γ' precipitates in Ni-based superalloys. These alloys quickly surpassed Fe- and Co-based superalloys, which were strengthened by carbides and solid solution strengthening.
Although Cr was great for protecting the alloys from oxidation and corrosion up to 700 °C, metallurgists began decreasing Cr in favor of Al, which had oxidation resistance (but not corrosion resistance!) at much higher temperatures. The lack of Cr caused issues with hot corrosion, so coatings needed to be developed.
Around 1950, vacuum melting became commercialized, which allowed metallurgists to create higher purity alloys with more precise composition.
In the 60s and 70s, metallurgists changed focus from alloy chemistry to alloy processing. Directional solidification was developed to allow columnar or even single-crystal turbine blades. Oxide dispersion strengthening could obtain very fine grains and superplasticity.
Ni-based superalloy phases
- Gamma (γ): This phase composes the matrix of Ni-based superalloy. It is a solid solution fcc austenitic phase of the alloying elements.[18][19] Alloying elements found in most commercial Ni-based alloys are, C, Cr, Mo, W, Nb, Fe, Ti, Al, V, and Ta. During the formation of these materials, as the Ni-alloys are cooled from the melt, carbides begin to precipitate, at even lower temperatures γ' phase precipitates.[19][20]
- Gamma prime (γ'): This phase constitutes the precipitate used to strengthen the alloy. It is an intermetallic phase based on Ni3(Ti,Al) which have an ordered FCC L12 structure.[18] The γ' phase is coherent with the matrix of the superalloy having a lattice parameter that varies by around 0.5%. Ni3(Ti,Al) are ordered systems with Ni atoms at the cube faces and either Al or Ti atoms at the cube edges. As particles of γ' precipitates aggregate, they decrease their energy states by aligning along the <100> directions forming cuboidal structures.[19] This phase has a window of instability between 600 °C and 850 °C, inside of which γ' will transform into the HCP η phase. For applications at temperatures below 650 °C, the γ" phase can be utilized for strengthening.[21]

- Gamma double prime (γ"): This phase typically possesses the composition of Ni3Nb or Ni3V and is used to strengthen Ni-based superalloys at lower temperatures (<650 °C) relative to γ'. The crystal structure of γ" is body-centered tetragonal (BCT), and the phase precipitates as 60 nm by 10 nm discs with the (001) planes in γ" parallel to the {001} family in γ. These anisotropic discs form as a result of lattice mismatch between the BCT precipitate and the FCC matrix. This lattice mismatch leads to high coherency strains which, together with order hardening, comprise the primary strengthening mechanisms. The γ" phase is unstable above approximately 650 °C.[21]
- Carbide phases: Carbide formation is usually considered deleterious although in Ni-based superalloys they are used to stabilize the structure of the material against deformation at high temperatures. Carbides form at the grain boundaries inhibiting grain boundary motion.[18][19]
- Topologically close-packed (TCP) phases: The term "TCP phase" refers to any member of a family of phases (including the σ phase, the χ phase, the μ phase, and the Laves phase) which are not atomically close-packed but possess some close-packed planes with HCP stacking. TCP phases are characterized by their tendency to be highly brittle and deplete the γ matrix of strengthening, solid solution refractory elements (including Cr, Co, W, and Mo). These phases form as a result of kinetics after long periods of time (thousands of hours) at high temperatures (>750 °C).[21]
Nickel-based superalloy MAR-M 247 had very good fatigue performance at temperatures of 800 and 900 °C.[22]
History and development of Co-based superalloys
Historically, Co-based superalloys have depended on carbide precipitation and solid solution strengthening for mechanical properties. While these strengthening mechanisms are inferior to gamma prime (γ') precipitation strengthening,[1] cobalt has a higher melting point than currently ubiquitous nickel-based superalloys and has superior hot corrosion resistance and thermal fatigue. As a result, carbide-strengthened Co-based superalloys are used in lower stress, higher temperature applications such as stationary vanes in gas turbines.[23]
However, recent research has shown that cobalt can exhibit the γ' phase. Actually, the first reported existence of γ' occurred in a 1971 PhD dissertation,[10] but was never published. The γ/γ' microstructure was rediscovered and first published in 2006 by Sato et al.[9] That γ' phase was Co3(Al, W). It was furthermore found that Mo, Ti, Nb, V, and Ta partition to the γ' phase, while Fe, Mn, and Cr partition to the matrix γ.
The next family of Co-based superalloys was discovered in 2015 by Makineni et al. This family has a similar γ/γ' microstructure, but is tungsten-free and has a γ' phase of Co3(Al,Mo,Nb).[12] Since tungsten is a very heavy element, the elimination of tungsten makes Co-based alloys increasingly viable in turbines for aircraft, where low density is especially important.
The most recently discovered family of superalloys was computationally predicted in a high throughput study by Nyshadham et al.[24] in 2017, and demonstrated in the lab by Reyes Tirado et al. in 2018.[15] This γ' phase is again tungsten free and has the composition Co3(Nb,V) and Co3(Ta,V).
Co-based superalloy phases
- Gamma (γ): Similar to Ni-based superalloys, this is the phase of the superalloy’s matrix. While not used commercially to the extent of Ni-based superalloys, alloying elements found in research Co-based alloys are C, Cr, W, Ni, Ti, Al, Ir, and Ta.[9][25] Chromium is also used in Cobalt based superalloys (occasionally up to 20 wt.%) as it provides oxidation and corrosion resistance, critical for material use in gas turbines.[26]
- Gamma Prime (γ'): Just as in Ni-based super alloys, this phase constitutes the precipitate used to strengthen the alloy. In this case, it is usually close packed with a L12 structure of Co3Ti or fcc Co3Ta, though both W and Al have been found to integrate into these cuboidal precipitates quite well. The elements Ta, Nb, and Ti integrate into the γ’ phase and are quite effective at stabilizing it at high temperatures. This stabilization is quite important as the lack of stability is one of the key factors that makes Co-based superalloys weaker than their Ni-base cousins at elevated temperatures.[9][27]
- Carbide Phases: As is common with carbide formation, its appearance in Co-based superalloys does provide precipitation hardening, but decreases low-temperature ductility.[25]
- Topologically Close-Packed (TCP) phases may also appear in some developmental Co-based superalloys, but a major point of engineering these alloys is avoiding TCPs.
Fe-based superalloy phases
The use of steels in superalloy applications is of interest because certain steel alloys have showed creep and oxidation resistance similar to that of Ni-based superalloys, while being far less expensive to produce.
Gamma (γ): Like the phases found in Ni-based superalloys, Fe-based alloys feature a matrix phase of austenite iron (FCC). Alloying elements that are commonly found in these stainless steel alloys include: Al, B, C, Co, Cr, Mo, Ni, Nb, Si, Ti, W, and Y.[28] While Al is introduced for its oxidation benefits, Al additions must be kept at low weight fractions (wt.%) because Al stabilizes a ferritic (BCC) primary phase matrix, which is an undesirable phase in superalloy microstructures, as it is inferior to the high temperature strength exhibited by an austenitic (FCC) primary phase matrix.[29]
Gamma-prime (γ’): This phase is introduced as precipitates to strengthen the alloy. Like in Ni-based alloys, γ’-Ni3Al precipitates can be introduced with the proper balance of Al, Ni, Nb, and Ti additions.
Microstructure of Fe-based superalloys
Two major types of austenitic stainless steels exist and are characterized by the oxide layer that forms at the surface of the steel: chromia-forming or alumina-forming stainless steel. Chromia-forming stainless steel is the most common type of stainless steel produced. However, chromia-forming steels do not exhibit high creep resistance at high operating temperatures, especially in environments with water vapor, when compared to Ni-based superalloys. Exposure to water vapor at high operating temperatures can result in an increase in internal oxidation in chromia-forming alloys and rapid formation of volatile Cr (oxy)hydroxides, both of which can reduce the durability and lifetime of the alloy.[29]
Alumina-forming austenitic stainless steels feature a single-phase matrix of austenite iron (FCC) with an alumina oxide at the surface of the steel. Alumina is more thermodynamically stable in oxygen than chromia. More commonly, however, precipitate phases are introduced to increase strength and creep resistance. In alumina-forming steels, NiAl precipitates are introduced to act as Al reservoirs to maintain the protective alumina layer. In addition, Nb and Cr additions help form and stabilize alumina by increasing precipitate volume fractions of NiAl.[29]
Research endeavors for the development of alumina-forming, Fe-base superalloys have shown at least 5 grades of alumina-forming austenitic (AFA) alloys, with different operating temperatures at oxidation in air + 10% water vapor:[30]
- AFA Grade: (50-60)Fe-(20-25)Ni-(14-15)Cr-(2.5-3.5)Al-(1-3)Nb wt.% base
- 750-800 °C operating temperatures at oxidation in air + 10% water vapor
- Low Nickel AFA Grade: 63Fe-12Ni-14Cr-2.5Al-0.6Nb-5Mn3Cu wt.% base
- 650 °C operating temperatures at oxidation in air + 10% water vapor
- High Performance AFA Grade: (45-55)Fe-(25-30)Ni-(14-15)Cr(3.5-4.5)Al-(1-3)Nb-(0.02-0.1)Hf/Y wt.% base
- 850-900 °C operating temperatures at oxidation in air + 10% water vapor
- Cast AFA Grade: (35-50)Fe-(25-35)Ni-14Cr-(3.5-4)Al-1Nb wt.% base
- 750-1100 °C operating temperatures at oxidation in air + 10% water vapor, depending upon Ni wt.%
- AFA superalloy (40-50)Fe-(30-35)Ni-(14-19)Cr-(2.5-3.5)Al-3Nb
- 750-850 °C operating temperatures at oxidation in air + 10% water vapor
Operating temperatures with oxidation in air and no water vapor are expected to be higher. In addition, an AFA superalloy grade was shown to exhibit a creep strength approaching that of the nickel-based alloy UNS N06617.
Microstructure of superalloys
In pure Ni3Al phase atoms of aluminium are placed at the vertices of the cubic cell and form the sublattice A. Atoms of nickel are located at centers of the faces and form the sublattice B. The phase is not strictly stoichiometric. There may exist an excess of vacancies in one of the sublattices, which leads to deviations from stoichiometry. Sublattices A and B of the γ'-phase can solute a considerable proportion of other elements. The alloying elements are dissolved in the γ-phase as well. The γ'-phase hardens the alloy through an unusual mechanism called the yield strength anomaly. Dislocations dissociate in the γ'-phase, leading to the formation of an anti-phase boundary. At elevated temperature, the free energy associated with the anti-phase boundary (APB) is considerably reduced if it lies on a particular plane, which by coincidence is not a permitted slip plane. One set of partial dislocations bounding the APB cross-slips so that the APB lies on the low-energy plane, and, since this low-energy plane is not a permitted slip plane, the dissociated dislocation is now effectively locked. By this mechanism, the yield strength of γ'-phase Ni3Al actually increases with temperature up to about 1000 °C, giving superalloys their currently unrivaled high-temperature strength.
Initial material selection for blade applications in gas turbine engines included alloys like the Nimonic series alloys in the 1940s.[4] The early Nimonic series incorporated γ' Ni3(Al,Ti) precipitates in a γ matrix, as well as various metal-carbon carbides (e.g. Cr23C6) at the grain boundaries[31] for additional grain boundary strength. Turbine blade components were forged until vacuum induction casting technologies were introduced in the 1950s.[4] This process significantly improved cleanliness, reduced defects, and increased the strength and temperature capability of the material.
Modern superalloys were developed in the 1980s. The first generation superalloys incorporated increased aluminium, titanium, tantalum, and niobium content in order to increase the γ' volume fraction in these alloys. Examples of first generation superalloys include: PWA1480, René N4 and SRR99. Additionally, the volume fraction of the γ' precipitates increased to about 50–70% with the advent of single crystal, or monocrystal, solidification techniques (see Bridgman technique) for superalloys that enable grain boundaries to be entirely eliminated from a casting. Because the material contained no grain boundaries, carbides were unnecessary as grain boundary strengthers and were thus eliminated.[4]
The second and third generation superalloys introduced about 3 and 6 weight per cent Rhenium, for increased temperature capability. Re is a slow diffuser and typically partitions to the γ matrix, decreasing the rate of diffusion (and thereby high temperature creep) and improving high temperature performance and increasing service temperatures by 30 °C and 60 °C in second and third generation superalloys, respectively.[32] Re has also been shown to promote the formation of rafts of the γ' phase (as opposed to cuboidal precipitates). The presence of rafts can decrease creep rate in the power-law regime (controlled by dislocation climb), but can also potentially increase the creep rate if the dominant mechanism is particle shearing. Furthermore, Re tends to promote the formation of brittle TCP phases, which has led to the strategy of reducing Co, W, Mo, and particularly Cr. Younger generations of Ni-based superalloys have significantly reduced Cr content for this reason, however with the reduction in Cr comes a reduction in oxidation resistance. Advanced coating techniques are now used to offset the loss of oxidation resistance accompanying the decreased Cr contents.[21][33] Examples of second generation superalloys include PWA1484, CMSX-4 and René N5. Third generation alloys include CMSX-10, and René N6. Fourth, Fifth, and even Sixth generation superalloys have been developed which incorporate ruthenium additions, making them more expensive still than the prior generation's Re-containing alloys. The effect of Ru on the promotion of TCP phases is not well-determined. Early reports determined that Ru decreased the supersaturation of Re in the matrix and thereby diminished the susceptibility to TCP phase formation.[34] More recent studies have noted the opposite effect. Chen, et al., found that in two alloys differing significantly only in Ru content (USTB-F3 and USTB-F6) that the addition of Ru increased both the partitioning ratio as well as the supersaturation in the γ matrix of Cr and Re, and thereby promoted the formation of TCP phases.[35]
The current trend is to avoid very expensive and very heavy elements. An example is Eglin steel, a budget material with compromised temperature range and chemical resistance. It does not contain rhenium or ruthenium and its nickel content is limited. To reduce fabrication costs, it was chemically designed to melt in a ladle (though with improved properties in a vacuum crucible). Also, conventional welding and casting is possible before heat-treatment. The original purpose was to produce high-performance, inexpensive bomb casings, but the material has proven widely applicable to structural applications, including armor.
Single-crystal superalloys
Single-crystal superalloys (SX or SC superalloys) are formed as a single crystal using a modified version of the directional solidification technique, so there are no grain boundaries in the material. The mechanical properties of most other alloys depend on the presence of grain boundaries, but at high temperatures, they would participate in creep and must be replaced by other mechanisms. In many such alloys, islands of an ordered intermetallic phase sit in a matrix of disordered phase, all with the same crystalline lattice. This approximates the dislocation-pinning behavior of grain boundaries, without introducing any amorphous solid into the structure.
Single crystal (SX) superalloys have wide application in the high-pressure turbine section of aero and industrial gas turbine engines due to the unique combination of properties and performance. Since introduction of single crystal casting technology, SX alloy development has focused on increased temperature capability, and major improvements in alloy performance have been associated with the introduction of new alloying elements, including rhenium (Re) and ruthenium (Ru).[36]
With increasing turbine entry temperature, it is important to gain a fundamental understanding of the physical phenomena occurring during creep deformation of single crystal superalloys under such extreme condition (i.e. high temperature and high stress). The creep deformation behavior of superalloy single crystal is strongly temperature, stress, orientation and alloy dependent. For a single-crystal superalloy, there are 3 different modes of creep deformation under regimes of different temperature and stress: Rafting, Tertiary and Primary.[37] At low temperature (~750 °C), SX alloys exhibits mostly primary creep behavior. Matan et al. concluded that the extent of primary creep deformation depends strongly on the angle between the tensile axis and the <001>/<011> symmetry boundary.[38] At temperature above 850 °C, tertiary creep dominates and promotes strain softening behavior.[4] When temperature exceeds 1000 °C, the rafting effect is prevalent where cubic particles transform into flat shapes under tensile stress[39] The rafts would also form perpendicular to the tensile axis, since γ phase was transported out of the vertical channels and into the horizontal ones. After conducting unaxial creep deformation of <001> orientated CMSX-4 single crystal superalloy at 1105 °C and 100 MPa, Reed et al. has established that rafting is beneficial to creep life since it delays evolution of creep strain. In addition, rafting would occur quickly and suppress the accumulation of creep strain until a critical strain is reached.[40]
Oxidation in superalloys
For superalloys operating at high temperatures and exposed to corrosive environments, the oxidation behavior is of paramount concern. Oxidation involves chemical reactions of the alloying elements with oxygen to form new oxide phases, generally at the surface of the metal. If unmitigated, oxidation can degrade the alloy over time in a variety of ways, including:[41][42]
- sequential oxidation, cracking, and spalling of the surface, leading to erosion of the alloy over time.
- embrittlement of the surface through the introduction of oxide phases, promoting crack formation and fatigue failure
- depletion of key alloying elements, affecting the mechanical properties of the superalloy and possibly compromising its performance.
The primary strategy used to limit these deleterious processes is called selective oxidation. Simply, the alloy is designed such that the ratio of alloying elements promotes formation of a specific oxide phase that can then act as a barrier to further oxidation. Most commonly, aluminum and chromium are used in this role, because they form relatively thin and continuous oxide layers of alumina (Al2O3) and chromia (Cr2O3), respectively. Furthermore, they possess low oxygen diffusivities, effectively halting further oxidation beneath this layer. In the ideal case, oxidation proceeds through 2 stages. First, transient oxidation involves the conversion of various elements, especially the majority elements (e.g. nickel or cobalt). Transient oxidation proceeds until the selective oxidation of the sacrificial element forms a complete barrier layer.[41]
The protective effect of selective oxidation can be undermined by numerous mechanisms. The continuity of the thin sacrificial oxide layer can be compromised by mechanical disruption due to stress or may be disrupted as a result of the kinetics of oxidation (e.g. if diffusion of oxygen is too fast). If the layer is not continuous, its effectiveness as a diffusion barrier to oxygen is significantly reduced. The stability of the oxide layer is also strongly influenced by the presence of other minority elements. For example, the addition of boron, silicon, and yttrium to superalloys promotes oxide layer adhesion, reducing spalling and maintaining the integrity of the protective oxide layer.[43]
Oxidation is only the most basic form of chemical degradation superalloys may experience. More complex corrosion processes are common when operating environments include salts and sulfur compounds, or under chemical conditions that change dramatically over time. These issues and those of basic oxidation are often also addressed through thin coatings.
Superalloy processing
The historical developments in superalloy processing have brought about considerable increases in superalloy operating temperatures. Superalloys were originally iron based and cold wrought prior to the 1940s. In the 1940s investment casting of cobalt base alloys significantly raised operating temperatures. The development of vacuum melting in the 1950s allowed for very fine control of the chemical composition of superalloys and reduction in contamination and in turn led to a revolution in processing techniques such as directional solidification of alloys and single crystal superalloys.[44]
There are many forms of superalloy present within a gas turbine engine, and processing methods vary widely depending on the necessary properties of each specific part.
Casting and forging
Casting and forging are traditional metallurgical processing techniques that can be used to generate both polycrystalline and monocrystalline products. Polycrystalline casts tend to have higher fracture resistance, while monocrystalline casts have higher creep resistance.
Jet turbine engines employ both poly and mono crystalline components to take advantage of their individual strengths. The disks of the high-pressure turbine, which are near the central hub of the engine are polycrystalline. The turbine blades, which extend radially into the engine housing, experience a much greater centripetal force, necessitating creep resistance. As a result, turbine blades are typically monocrystalline or polycrystalline with a preferred crystal orientation.
Investment casting
Investment casting is a metallurgical processing technique in which a wax form is fabricated and used as a template for a ceramic mold. Briefly, a ceramic mold is poured around the wax form, the wax form is melted out of the ceramic mold, and molten metal is poured into the void left by the wax. This leads to a metal form in the same shape as the original wax form. Investment casting leads to a polycrystalline final product, as nucleation and growth of crystal grains occurs at numerous locations throughout the solid matrix. Generally, the polycrystalline product has no preferred grain orientation.
Directional solidification
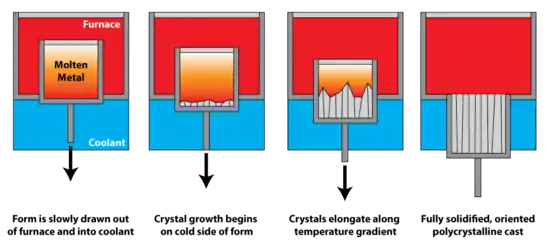
Directional solidification uses a thermal gradient to promote nucleation of metal grains on a low temperature surface, as well as to promote their growth along the temperature gradient. This leads to grains elongated along the temperature gradient, and significantly greater creep resistance parallel to the long grain direction. In polycrystalline turbine blades, directional solidification is used to orient the grains parallel to the centripetal force. It is also known as dendritic solidification.
Single crystal growth
Single crystal growth starts with a seed crystal which is used to template growth of a larger crystal. The overall process is lengthy, and additional processing via machining is necessary after the single crystal is grown.
Powder metallurgy
Powder metallurgy is a class of modern processing techniques in which metals are first converted into a powdered form, and then formed into the desired shape by heating below the melting point. This is in contrast to casting, which occurs with molten metal. Superalloy manufacturing often employs powder metallurgy because of its material efficiency - typically much less waste metal must be machined away from the final product—and its ability to facilitate mechanical alloying. Mechanical alloying is a process by which reinforcing particles are incorporated into the superalloy matrix material by repeated fracture and welding.[45]
Sintering and hot isostatic pressing
Sintering and hot isostatic pressing are processing techniques used to densify materials from a loosely packed "green body" into a solid object with physically merged grains. Sintering occurs below the melting point, and causes adjacent particles to merge at their boundaries, leading to a strong bond between them. In hot isostatic pressing, a sintered material is placed in a pressure vessel and compressed from all directions (isostatically) in an inert atmosphere to affect densification.[46]
Additive manufacturing
Selective laser melting (also known as powder bed fusion) is an additive manufacturing procedure used to create intricately detailed forms from a CAD file. In CAD, a shape is designed and then converted into slices. These slices are sent to a laser writer to print the final product. In brief, a bed of metal powder is prepared, and the first slice of the CAD design is formed in the powder bed by a high energy laser sintering the particles together. After this first slice is generated, the powder bed moves downwards, and a new batch of metal powder is rolled over the top of the slice. The second layer is then sintered with the laser, and the process is repeated until all the slices in the CAD file have been processed.[47] Due to the nature of many additive manufacturing processes, porosity can be present in products made by selective laser melting. Many products will often undergo a heat treatment or hot isostatic pressing procedure to densify the product and reduce porosity which can result in cracking.[48]
Coating of superalloys
In modern gas turbine, the turbine entry temperature (~1750K) has exceeded the incipient melting temperature of superalloys (~1600K), with the help of surface engineering. Under such extreme working condition, the qualification of coating becomes vital.[49]
Different types of coating
Historically, three "generations" of coatings have been developed: diffusion coatings, overlay coatings and thermal barrier coatings. Diffusion coatings, mainly constituted with aluminide or platinum-aluminide, is still the most common form of surface protection. To further enhance resistance to corrosion and oxidation, MCrAlX-based overlay coatings (M=Ni or Co, X=Y, Hf, Si) are deposited to surface of superalloys. Compared to diffusion coatings, overlay coatings are less dependent on the composition of the substrate, but also more expensive, since they must be carried out by air or vacuum plasma spraying (APS/VPS)[50] or else electron beam physical vapour deposition (EB-PVD).[51] Thermal barrier coatings provide by far the best enhancement in working temperature and coating life. It is estimated that modern TBC of thickness 300 μm, if used in conjunction with a hollow component and cooling air, has the potential to lower metal surface temperatures by a few hundred degrees.[52]
Thermal barrier coatings
Thermal barrier coatings (TBCs) are used extensively on the surface of superalloy in both commercial and military gas turbine engines to increase component life and engine performance.[53] A coating of about 1-200 µm can reduce the temperature at the superalloy surface by up to 200K. TBCs are really a system of coatings consisting of a bond coat, a thermally grown oxide (TGO), and a thermally insulating ceramic top coat. In most applications, the bond coat is either a MCrAlY (where M=Ni or NiCo) or a Pt modified aluminide coating. A dense bond coat is required to provide protection of the superalloy substrate from oxidation and hot corrosion attack and to form an adherent, slow growing TGO on its surface. The TGO is formed by oxidation of the aluminum that is contained in the bond coat. The current (first generation) thermal insulation layer is composed of 7wt % yttria-stabilized zirconia (7YSZ) with a typical thickness of 100–300 µm. Yttria stabilized zirconia is used due to its low thermal conductivity (2.6W/mK for fully dense material), relatively high coefficient of thermal expansion, and good high temperature stability. The electron beam directed vapor deposition (EB-DVD) process used to apply the TBC to turbine airfoils produces a columnar microstructure with several levels of porosity. The porosity between the columns is critical to providing strain tolerance (via a very low in-plane modulus), as it would otherwise spall on thermal cycling due to thermal expansion mismatch with the superalloy substrate. The porosity within the columns reduces the thermal conductivity of the coating.
Bond coat
The bond coat adheres the thermal barrier coating to the superalloy substrate. Additionally, the bond coat provides oxidation protection and functions as a diffusion barrier against the motion of substrate atoms towards the environment. There are five major types of bond coats, the aluminides, the platinum-aluminides, MCrAlY, cobalt-cermets, and nickel-chromium. For the aluminide bond coatings, the final composition and structure of the coating depends on the composition of the substrate. Aluminides also lack ductility below 750 °C, and exhibit a limited by thermomechanical fatigue strength. The Pt-aluminides are very similar to the aluminide bond coats except for a layer of Pt (5—10 μm) deposited on the blade. The Pt is believed to aid in oxide adhesion and contributes to hot corrosion. The cost of Pt plating is justified by the increased blade life span. The MCrAlY is the latest generation of bond coat and does not strongly interact with the substrate. Normally applied by plasma spraying, MCrAlY coatings are secondary aluminum oxide formers. This means that the coatings form an outer layer of chromium oxide (chromia), and a secondary aluminum oxide (alumina) layer underneath. These oxide formations occur at high temperatures in the range of those that superalloys usually encounter.[54] The chromia provides oxidation and hot-corrosion resistance. The alumina controls oxidation mechanisms by limiting oxide growth by self-passivating. The yttrium enhances the oxide adherence to the substrate, and limits the growth of grain boundaries (which can lead to flaking of the coating).[55] Investigation indicates that addition of rhenium and tantalum increases oxidation resistance. Cobalt-cermet based coatings consisting of materials such as tungsten carbide/cobalt can be used due to excellent resistance to abrasion, corrosion, erosion, and heat.[56] These cermet coatings perform well in situations where temperature and oxidation damage are significant concerns, such as boilers. One of the unique advantages of cobalt cermet coatings is a minimal loss of coating mass over time, due to the strength of carbides within the mixture. Overall, cermet coatings are useful in situations where mechanical demands are equal to chemical demands for superalloys. Nickel-chromium coatings are used most frequently in boilers fed by fossil fuels, electric furnaces, and waste incineration furnaces, where the danger of oxidizing agents and corrosive compounds in the vapor must be dealt with.[57] The specific method of spray-coating depends on the composition of the coatings. Nickel-chromium coatings that also contain iron or aluminum perform much better (in terms of corrosion resistance) when they are sprayed and laser glazed, while pure nickel-chromium coatings perform better when thermally sprayed exclusively.[58]
Process methods of coating
Superalloy products that are subjected to high working temperatures and corrosive atmosphere (such as high-pressure turbine region of jet engines) are coated with various kinds of coating. Several kinds of coating process are applied: pack cementation process, gas phase coating (both are a type of chemical vapor deposition (CVD)), thermal spraying, and physical vapor deposition. In most cases, after the coating process near-surface regions of parts are enriched with aluminium, the matrix of the coating being nickel aluminide.
Pack cementation process
Pack cementation is a widely used chemical vapor deposition technique which consists of immersing the components to be coated in a metal powder mixture and ammonium halide activators and sealing them in a retort. The entire apparatus is placed inside a furnace and heated in a protective atmosphere to a lower than normal temperature for diffusion to take place, due to the halide salts chemical reaction which causes an eutectic bond between the two metals. The new surface alloy that is formed due to thermal diffused ion migration has a metallurgical bond to the surface substrate and a true intermetallic layer found in the gamma layer of the new surface alloys.
The traditional pack consists of four components:
Substrate or parts- Ferrous and non-ferrous Powdered alloy- (Ti and/or Al, Si and/or Zn, B and/ or Cr) Halide salt activator- Ammonium halide salts Relatively inert filler powder (Al2O3, SiO2, or SiC) Temperatures below (750 °C) This process includes but is not limited to:
Aluminizing Chromizing Siliconizing Sherardizing Boronizing Titaniumizing
Pack Cementation has had a revival in the last 10 years as it is being combined with other chemical processes to lower the temperatures of metal combinations even further and give intermetallic properties to different alloy combinations for surface treatments.
Thermal spraying
Thermal spraying is a process of applying coatings by heating a feedstock of precursor material and spraying it on a surface. Different specific techniques are used depending on desired particle size, coat thickness, spray speed, desired area, etc.[59] The coatings applied by thermal spraying of any kind, however, rely on adhesion to the surface. As a result, the surface of the superalloy must be cleaned and prepared, usually polished, before application of the thermal coating.[60]
Plasma spraying
Of the various thermal spray methods, one of the more ideal and commonly used techniques for coating superalloys is plasma spraying. This is due to the versatility of usable coatings, and the high-temperature performance of plasma-sprayed coatings.[61] Plasma spraying can accommodate a very wide range of materials, much more so than other techniques. As long as the difference between melting and decomposition temperatures is greater than 300 Kelvin, a material can be melted and applied as a coating via plasma spraying.[62]
Gas phase coating
This process is carried out at higher temperatures, about 1080 °C. The coating material is usually loaded onto special trays without physical contact with the parts to be coated. The coating mixture contains active coating material and activator, but usually does not contain thermal ballast. As in the pack cementation process, the gaseous aluminium chloride (or fluoride) is transferred to the surface of the part. However, in this case the diffusion is outwards. This kind of coating also requires diffusion heat treatment.
Failure mechanisms in thermal barrier coating systems
Failure of thermal barrier coating usually manifests as delamination, which arises from the temperature gradient during thermal cycling between ambient temperature and working conditions coupled with the difference in thermal expansion coefficient of the substrate and coating. It is rare for the coating to fail completely – some pieces of it remain intact, and significant scatter is observed in the time to failure if testing is repeated under identical conditions.[4] There are various degradation mechanisms for thermal barrier coating,[63][64] and some or all of these must operate before failure finally occurs:
- Oxidation at the interface of thermal barrier coating and underlying bond coat;[65]
- The depletion of aluminum in bond coat due to oxidation[66] and diffusion with substrate;[67]
- Thermal stresses from mismatch in thermal expansion coefficient and growth stress due to the formation of thermally grown oxide layer;[68]
- Imperfections near thermally grown oxide layer;[69][70][71]
- Various other complicating factors during engine operation.[72][73][74][75][76]
Additionally, TBC life is very dependent upon the combination of materials (substrate, bond coat, ceramic) and processes (EB-PVD, plasma spraying) used.
Applications
Nickel-based superalloys are used in load-bearing structures to the highest homologous temperature of any common alloy system (Tm = 0.9, or 90% of their melting point). Among the most demanding applications for a structural material are those in the hot sections of turbine engines. The preeminence of superalloys is reflected in the fact that they currently comprise over 50% of the weight of advanced aircraft engines. The widespread use of superalloys in turbine engines coupled with the fact that the thermodynamic efficiency of turbine engines is increased with increasing turbine inlet temperatures has, in part, provided the motivation for increasing the maximum-use temperature of superalloys. In fact, during the past 30 years turbine airfoil temperature capability has increased on average by about 4 °F (2.2 °C) per year. Two major factors which have made this increase possible are
- Advanced processing techniques, which improved alloy cleanliness (thus improving reliability) and/or enabled the production of tailored microstructures such as directionally solidified or single-crystal material.
- Alloy development resulting in higher-use-temperature materials primarily through the additions of refractory elements such as Re, W, Ta, and Mo.
About 60% of the use-temperature increases have occurred due to advanced cooling concepts; 40% have resulted from material improvements. State-of-the-art turbine blade surface temperatures are near 2,100 °F (1,150 °C); the most severe combinations of stress and temperature corresponds to an average bulk metal temperature approaching 1,830 °F (1,000 °C).
Although Nickel-based superalloys retain significant strength to temperatures near 1,800 °F (980 °C), they tend to be susceptible to environmental attack because of the presence of reactive alloying elements (which provide their high-temperature strength). Surface attack includes oxidation, hot corrosion, and thermal fatigue. In the most demanding applications, such as turbine blade and vanes, superalloys are often coated to improve environmental resistance.[18]
In general, high temperature materials are needed for energy conversion and energy production applications. Maximum energy conversion efficiency is desired in these energy applications, which can be achieved by increasing operating temperatures, as described by the Carnot cycle. Because Carnot efficiency is limited by the temperature difference between the hot and cold reservoirs, higher operating temperatures result in higher energy conversion efficiencies. Operating temperatures are limited by the performance of today’s superalloys, and currently, most applications operate at around 1000 °C-1400 °C. Energy applications and their superalloy components include:[77]
- Gas turbines (turbine blades)
- Solar thermal power plants (stainless steel rods containing heated water)
- Steam turbines (turbine blades and boiler housing)
- Heat exchangers for nuclear reactor systems
Alumina-forming stainless steels can be processed via melting and ladle casting, similar to the production of more common steels. Compared to vacuum casting processes, ladle casting is much less expensive. In addition, alumina-forming stainless steel has been shown to be weldable and has potential for use in high performance automotive applications, such as for high temperature exhaust piping and in heat capture and reuse.
Research and development of new superalloys
The availability of superalloys during past decades has led to a steady increase in turbine entry temperatures, and the trend is expected to continue. Sandia National Laboratories is studying a new method for making superalloys, known as radiolysis. It introduces an entirely new area of research into creating alloys and superalloys through nanoparticle synthesis. This process holds promise as a universal method of nanoparticle formation. By developing an understanding of the basic material science behind these nanoparticle formations, there is speculation that it might be possible to expand research into other aspects of superalloys.
There may be considerable disadvantages in making alloys by this method. About half of the use of superalloys is in applications where the service temperature is close to the melting temperature of the alloy. It is common therefore to use single crystals. The above method produces polycrystalline alloys, which suffer from an unacceptable level of creep.
Future paradigms in alloy development are expected to focus on weight reduction and improving oxidation and corrosion resistance while maintaining the strength of the alloy. Furthermore, with the increasing demand for turbine blades for power generation, another focus of alloy design is to reduce the cost of superalloys.
There has been ongoing research and development of new stainless steel alloys because of the lower costs in producing such alloys, as well as the need for an austenitic stainless steel with high-temperature corrosion resistance in environments with water vapor. Research is focusing on increasing high-temperature tensile strength, toughness, and creep resistance to compete with Ni-based superalloys.[30]
A new class of alumina-forming austenitic stainless steel is actively being developed for use in high-temperature applications by Oak Ridge National Laboratory. Initial research showed similar creep and corrosion resistance at 800 °C to that of other austenitic alloys, including Ni-based superalloys.[30]
Development of AFA superalloys with a 35 wt.% Ni-base have shown potential for use in operating temperatures upwards to 1,100 °C.[30]
References
- Sims, C.T. (1984). "A History of Superalloy Metallurgy for Superalloy Metallurgists". Superalloys 1984 (Fifth International Symposium). pp. 399–419. doi:10.7449/1984/Superalloys_1984_399_419.
- Carter, Tim J (April 2005). "Common failures in gas turbine blades". Engineering Failure Analysis. 12 (2): 237–247. doi:10.1016/j.engfailanal.2004.07.004.
- Sims, C.T. (1984). "A History of Superalloy Metallurgy for Superalloy Metallurgists". Superalloys 1984 (Fifth International Symposium). pp. 399–419. doi:10.7449/1984/Superalloys_1984_399_419.
- Reed, R. C (2008). The Superalloys: Fundamentals and Applications. Cambridge: Cambridge University Press. ISBN 9780521070119.
- Klein, L.; Shen, Y.; Killian, M. S.; Virtanen, S. (2011). "Effect of B and Cr on the high temperature oxidation behaviour of novel γ/γ′-strengthened Co-base superalloys". Corrosion Science. 53 (9): 2713–720. doi:10.1016/j.corsci.2011.04.020.
- Shinagawa, K.; Omori, Toshihiro; Oikawa, Katsunari; Kainuma, Ryosuke; Ishida, Kiyohito (2009). "Ductility Enhancement by Boron Addition in Co–Al–W High-temperature Alloys". Scripta Materialia. 61 (6): 612–15. doi:10.1016/j.scriptamat.2009.05.037.
- Giamei, Anthony (September 2013). "Development of Single Crystal Superalloys: A Brief History". Advanced Materials & Processes: 26–30 – via asminternational.
- Akca, Enes; Gursel, Ali (2015). "A Review on Superalloys and IN718 Nickel-Based INCONEL Superalloy". Periodicals of Engineering and Natural Sciences. 3 (1): 15–27 – via pen.ius.edu.ba.
- Sato, J (2006). "Cobalt-Base High-Temperature Alloys". Science. 312 (5770): 90–91. Bibcode:2006Sci...312...90S. doi:10.1126/science.1121738. PMID 16601187. S2CID 23877638.
- Lee, C. S. (1971). Precipitation-hardening characteristics of ternary cobalt - aluminum - X alloys (PhD dissertation). University of Arizona.
- Suzuki, A.; DeNolf, Garret C.; Pollock, Tresa M. (2007). "Flow Stress Anomalies in γ/γ′ Two-phase Co–Al–W-base Alloys". Scripta Materialia. 56 (5): 385–88. doi:10.1016/j.scriptamat.2006.10.039.
- Makineni, S. K.; Nithin, B.; Chattopadhyay, K. (March 2015). "A new tungsten-free γ–γ' Co–Al–Mo–Nb-based superalloy". Scripta Materialia. 98: 36–39. doi:10.1016/j.scriptamat.2014.11.009.
- Makineni, S. K.; Nithin, B.; Chattopadhyay, K. (February 2015). "Synthesis of a new tungsten-free γ–γ′ cobalt-based superalloy by tuning alloying additions". Acta Materialia. 85: 85–94. doi:10.1016/j.actamat.2014.11.016.
- Makineni, S. K.; Samanta, A.; Rojhirunsakool, T.; Alam, T.; Nithin, B.; Singh, A.K.; Banerjee, R.; Chattopadhyay, K. (September 2015). "A new class of high strength high temperature Cobalt based γ–γ′ Co–Mo–Al alloys stabilized with Ta addition". Acta Materialia. 97: 29–40. doi:10.1016/j.actamat.2015.06.034.
- Reyes Tirado, Fernando L.; Perrin Toinin, Jacques; Dunand, David C. (June 2018). "γ+γ′ microstructures in the Co-Ta-V and Co-Nb-V ternary systems". Acta Materialia. 151: 137–148. doi:10.1016/j.actamat.2018.03.057.
- Belan, Juraj (2016). "GCP and TCP Phases Presented in Nickel-base Superalloys". Materials Today: Proceedings. 3 (4): 936–941. doi:10.1016/j.matpr.2016.03.024.
- Rae, C.M.F.; Karunaratne, M.S.A.; Small, C.J.; Broomfield, R.W.; Jones, C.N.; Reed, R.C. (2000). "Topologically Close Packed Phases in an Experimental Rhenium-Containing Single Crystal Superalloy". Superalloys 2000 (Ninth International Symposium). pp. 767–776. doi:10.7449/2000/Superalloys_2000_767_776. ISBN 0-87339-477-1.
- Randy Bowman. "Superalloys: A Primer and History". Retrieved 6 March 2020 – via tms.org.
- Sabol, G. P.; Stickler, R. (1969). "Microstructure of Nickel-Based Superalloys". Physica Status Solidi (B). 35 (1): 11–52. Bibcode:1969PSSBR..35...11S. doi:10.1002/pssb.19690350102.
- Doi, M.; Miki, D.; Moritani, T.; Kozakai, T. (2004). "Gamma/Gamma-Prime Microstructure Formed by Phased Separation of Gamma-Prime Precipitates in a Ni-Al-Ti Alloy". Superalloys 2004 (Tenth International Symposium). pp. 109–114. doi:10.7449/2004/Superalloys_2004_109_114. ISBN 0-87339-576-X.
- Dunand, David C. "Materials Science & Engineering 435: High Temperature Materials". Northwestern University, Evanston. 25 February 2016. Lecture.
- Šmíd, Miroslav; Kunz, Ludvík; Hutař, Pavel; Hrbáček, Karel (1 January 2014). "High Cycle Fatigue of Nickel-based Superalloy MAR-M 247 at High Temperatures". Procedia Engineering. 74: 329–332. doi:10.1016/j.proeng.2014.06.273.
- Institute, Cobalt (14 February 2018). "Superalloys". www.cobaltinstitute.org. Retrieved 10 December 2019.
- Nyshadham, Chandramouli; Oses, Corey; Hansen, Jacob E.; Takeuchi, Ichiro; Curtarolo, Stefano; Hart, Gus L.W. (January 2017). "A computational high-throughput search for new ternary superalloys". Acta Materialia. 122: 438–447. doi:10.1016/j.actamat.2016.09.017. S2CID 11222811.
- Cui, C (2006). "A New Co-Base Superalloy Strengthened by γ' Phase". Materials Transactions. 47 (8): 2099–2102. doi:10.2320/matertrans.47.2099.
- Coutsouradis, D.; Davin, A.; Lamberigts, M. (April 1987). "Cobalt-based superalloys for applications in gas turbines". Materials Science and Engineering. 88: 11–19. doi:10.1016/0025-5416(87)90061-9.
- Suzuki, A.; Pollock, Tresa M. (2008). "High-temperature strength and deformation of γ/γ′ two-phase Co–Al–W-base alloys". Acta Materialia. 56 (6): 1288–97. doi:10.1016/j.actamat.2007.11.014.
- "Review: precipitation in austenitic stainless steels". www.phase-trans.msm.cam.ac.uk. Retrieved 2 March 2018.
- Brady, M. P.; Yamamoto, Y.; Santella, M. L.; Maziasz, P. J.; Pint, B. A.; Liu, C. T.; Lu, Z. P.; Bei, H. (July 2008). "The development of alumina-forming austenitic stainless steels for high-temperature structural use". JOM. 60 (7): 12–18. Bibcode:2008JOM....60g..12B. doi:10.1007/s11837-008-0083-2. S2CID 137354503.
- Muralidharan, G.; Yamamoto, Y.; Brady, M. P.; Walker, L. R.; Meyer III, H. M.; Leonard, D. N. (November 2016). "Development of Cast Alumina-Forming Austenitic Stainless Steels". JOM. 68 (11): 2803–2810. Bibcode:2016JOM....68k2803M. doi:10.1007/s11837-016-2094-8. OSTI 1362187. S2CID 137160315.
- Bombač, D.; Fazarinc, M.; Kugler, G.; Spajić, S. (2008). "Microstructure development of Nimonic 80A superalloys during hot deformation". Materials and Geoenvironment. 55 (3): 319–328. Retrieved 8 March 2020 – via ResearchGate.
- Reed, R. C (2006). The Superalloys: Fundamentals and Applications. Cambridge: Cambridge University Press. p. 121. ISBN 9780521070119.
- Dunand, David C. "High-Temperature Materials for Energy Conversion" Materials Science & Engineering 381: Materials for Energy-Efficient Technology. Northwestern University, Evanston. 3 February 2015. Lecture.
- O'Hara, K. S., Walston, W. S., Ross, E. W., Darolia, R. US Patent 5482789, 1996.
- Chen, J. Y.; Feng, Q.; Sun, Z. Q. (October 2010). "Topologically close-packed phase promotion in a Ru-containing single crystal superalloy". Scripta Materialia. 63 (8): 795–798. doi:10.1016/j.scriptamat.2010.06.019.
- Wahl, Jacqueline; Harris, Ken (2014). "New single crystal superalloys – overview and update". MATEC Web of Conferences. 14: 17002. doi:10.1051/matecconf/20141417002.
- Nabarro, F. R. N.; de Villiers, H. L. (1995). The Physics of creep : creep and creep-resistant alloys. London: Talylor and Francis. ISBN 9780850668520.
- Matan, N.; Cox, D. C.; Carter, P.; Rist, M. A.; Rae, C. M. F.; Reed, R. C. (1999). "Creep of CMSX-4 superalloy single crystals: effects of misorientation and temperature". Acta Materialia. 47 (5): 1549–1563. doi:10.1016/s1359-6454(99)00029-4.
- Nabarro, Frank R. N. (1996). "Rafting in Superalloys". Metallurgical and Materials Transactions A. 27 (3): 513–530. Bibcode:1996MMTA...27..513N. doi:10.1007/BF02648942. S2CID 137172614.
- Reed, R. C.; Matan, N.; Cox, D. C.; Rist, M. A.; Rae, C. M. F. (1999). "Creep of CMSX-4 superalloy single crystals: effects of rafting at high temperature". Acta Materialia. 47 (12): 3367–3381. doi:10.1016/S1359-6454(99)00217-7.
- Pettit, F.S.; Meier, G.H. (1984). "Oxidation and Hot Corrosion of Superalloys". Superalloys 1984 (Fifth International Symposium). pp. 651–687. doi:10.7449/1984/Superalloys_1984_651_687.
- Lund and Wagner. "Oxidation of Nickel- and Cobalt-Base Superalloys". DMIC report 214. 1 March 1965. Defense Metals Information Center, Batelle Memorial Institute, Columbus, Ohio.
- Klein, L.; Bauer, S.; Neumeier, S.; Göken, M.; Virtanan, S. (2011). "High temperature oxidation of γ/γ'-strengthened Co-based superalloys". Corrosion Science. 53 (5): 2027–2034. doi:10.1016/j.corsci.2011.02.033.
- C. Sims, N. Stoloff, W. Hagel, Superalloys II: High Temperature Materials for Aerospace and Industrial Power, 1987, John Wiley & Sons
- "PIM International Vol. 7 No. 1 March 2013". Powder Injection Moulding International. Retrieved 1 March 2016.
- Atkinson, H. V.; Davies, S. (December 2000). "Fundamental aspects of hot isostatic pressing: An overview". Metallurgical and Materials Transactions A. 31 (12): 2981–3000. Bibcode:2000MMTA...31.2981A. doi:10.1007/s11661-000-0078-2. S2CID 137660703.
- Gu, D D; Meiners, W; Wissenbach, K; Poprawe, R (May 2012). "Laser additive manufacturing of metallic components: materials, processes and mechanisms". International Materials Reviews. 57 (3): 133–164. doi:10.1179/1743280411Y.0000000014. S2CID 137144519.
- Graybill, Benjamin; Li, Ming; Malawey, David; Ma, Chao; Alvarado-Orozco, Juan-Manuel; Martinez-Franco, Enrique (18 June 2018). "Additive Manufacturing of Nickel-Based Superalloys". Volume 1: Additive Manufacturing; Bio and Sustainable Manufacturing. College Station, Texas, USA: American Society of Mechanical Engineers. doi:10.1115/MSEC2018-6666. ISBN 978-0-7918-5135-7.
- Y. Tamarin, Protective Coatings for Turbine Blades (Materials Park, OH: ASM International, 2002).
- J. R. Davis, ed., Handbook of Thermal Spray Technology (Materials Park, OH: The ASM Thermal Spray Society, 2004).
- Boone, D. H. (1986). "Physical vapour deposition processes". Materials Science and Technology. 2 (3): 220–224. doi:10.1179/mst.1986.2.3.220.
- Clarke, David R. (January 2003). "Materials selection guidelines for low thermal conductivity thermal barrier coatings". Surface and Coatings Technology. 163–164: 67–74. doi:10.1016/S0257-8972(02)00593-5.
- "Wadley Research Group '". University of Virginia. Retrieved 3 March 2016.
- Warnes, Bruce Michael (January 2003). "Improved aluminide/MCrAlX coating systems for super alloys using CVD low activity aluminizing". Surface and Coatings Technology. 163–164: 106–111. doi:10.1016/S0257-8972(02)00602-3.
- Tawancy, H.M.; Abbas, N.M.; Bennett, A. (December 1994). "Role of Y during high temperature oxidation of an M-Cr-Al-Y coating on an Ni-base superalloy". Surface and Coatings Technology. 68–69: 10–16. doi:10.1016/0257-8972(94)90130-9.
- D. Chuanxian; H. Bingtang; L. Huiling (24 August 1984). "Plasma-sprayed wear-resistant ceramic and cermet coating materials". Thin Solid Films. 118 (4): 485–493. Bibcode:1984TSF...118..485C. doi:10.1016/0040-6090(84)90277-3.
- Kawahara, Yuuzou (January 1997). "Development and application of high-temperature corrosion-resistant materials and coatings for advanced waste-to-energy plants". Materials at High Temperatures. 14 (3): 261–268. doi:10.1080/09603409.1997.11689552.
- Longa, Y.; Takemoto, M. (July 1992). "High-Temperature Corrosion of Laser-Glazed Alloys in Na 2 SO 4 -V 2 O 5". Corrosion. 48 (7): 599–607. doi:10.5006/1.3315978.
- G. R. Heath, P. Heimgartner, G. Irons, R. Miller, S. Gustafsson, Materials Science Forum 1997, 251–54, 809
- Knotek, O. (2001). "Thermal Spraying and Detonation Gun Processes" (PDF). In Bunshah, R. F. (ed.). Handbook of Hard Coatings: Deposition Technologies, Properties and Applications. Park Ridge, NJ: Noyes Pub.; Norwich, NY: William Andrew Pub. pp. 77–107. ISBN 9780815514381.
- Niranatlumpong, P.; Ponton, C. B.; Evans, H. E. (2000). "The Failure of Protective Oxides on Plasma-Sprayed NiCrAlY Overlay Coatings". Oxidation of Metals. 53 (3–4): 241–258. doi:10.1023/A:1004549219013. S2CID 136826569.
- P. Fauchais, A. Vardelle, M. Vardelle, Modelling of Plasma Spraying of Ceramic Films and Coatings, Ed. Vinenzini, Pub. Elsevier State Publishers B.V 1991.
- Evans, A. G.; Mumm, D. R.; Hutchinson, J. W.; Meier, G. H.; Pettit, F. S. (2001). "Mechanisms controlling the durability of thermal barrier coatings". Progress in Materials Science. 46 (5): 505–553. doi:10.1016/s0079-6425(00)00020-7.
- Wright, P. K.; Evans, A. G. (1999). "Mechanisms governing the performance of thermal barrier coatings". Current Opinion in Solid State and Materials Science. 4 (3): 255–265. Bibcode:1999COSSM...4..255W. doi:10.1016/s1359-0286(99)00024-8.
- Wright, P. K. (1998). "Influence of cyclic strain on life of a PVD TBC". Materials Science and Engineering. A245 (2): 191–200. doi:10.1016/S0921-5093(97)00850-2.
- Pint, B.A. (November 2004). "The role of chemical composition on the oxidation performance of aluminide coatings". Surface and Coatings Technology. 188–189: 71–78. doi:10.1016/j.surfcoat.2004.08.007.
- Baufeld, B.; Bartsch, M.; Broz, P.; Schmucker, M. (2004). "Microstructural changes as postmortem temperature indicator in Ni-Co-Cr-Al-Y oxidation protection coatings". Materials Science and Engineering. 384 (1–2): 162–171. doi:10.1016/j.msea.2004.05.052.
- Nychka, J.A; Clarke, D.R (September 2001). "Damage quantification in TBCs by photo-stimulated luminescence spectroscopy". Surface and Coatings Technology. 146–147: 110–116. doi:10.1016/S0257-8972(01)01455-4.
- Mumm, D. R.; Evans, A. G.; Spitsberg, I. T. (2001). "Characterisation of a cyclic displacement instability for a thermally grown oxide in a thermal barrier coating system". Acta Materialia. 49 (12): 2329–2340. doi:10.1016/s1359-6454(01)00071-4.
- Mumm, D. R.; Evans, A. G. (2000). "On the role of imperfections in the failure of a thermal barrier coating made by electron beam deposition". Acta Materialia. 48 (8): 1815–1827. doi:10.1016/s1359-6454(99)00473-5.
- Gell, M.; Vaidyanathan, K.; Barber, B.; Cheng, J.; Jordan, E. (1999). "Mechanism of spallation in platinum aluminide/electron beam physical vapor-deposited thermal barrier coatings". Metallurgical and Materials Transactions A. 30 (2): 427–435. Bibcode:1999MMTA...30..427G. doi:10.1007/s11661-999-0332-1. S2CID 137312835.
- Evans, A.G.; He, M.Y.; Hutchinson, J.W. (January 2001). "Mechanics-based scaling laws for the durability of thermal barrier coatings". Progress in Materials Science. 46 (3–4): 249–271. doi:10.1016/S0079-6425(00)00007-4.
- Schulz, U; Menzebach, M; Leyens, C; Yang, Y.Q (September 2001). "Influence of substrate material on oxidation behavior and cyclic lifetime of EB-PVD TBC systems". Surface and Coatings Technology. 146–147: 117–123. doi:10.1016/S0257-8972(01)01481-5.
- Chen, X; Wang, R; Yao, N; Evans, A.G; Hutchinson, J.W; Bruce, R.W (July 2003). "Foreign object damage in a thermal barrier system: mechanisms and simulations". Materials Science and Engineering: A. 352 (1–2): 221–231. doi:10.1016/S0921-5093(02)00905-X.
- Walston, W.S. (2004). "Coating and Surface Technologies for Turbine Airfoils". Superalloys 2004 (Tenth International Symposium). pp. 579–588. doi:10.7449/2004/Superalloys_2004_579_588. ISBN 0-87339-576-X.
- Mumm, D. R.; Watanabe, M.; Evans, A. G.; Pfaendtner, J. A. (2004). "The influence of test method on failure mechanisms and durability of a thermal barrier system". Acta Materialia. 52 (5): 1123–1131. CiteSeerX 10.1.1.514.3611. doi:10.1016/j.actamat.2003.10.045.
- Brady, M. P.; Muralidharan, G.; Leonard, D. N.; Haynes, J. A.; Weldon, R. G.; England, R. D. (December 2014). "Long-Term Oxidation of Candidate Cast Iron and Stainless Steel Exhaust System Alloys from 650 to 800 °C in Air with Water Vapor". Oxidation of Metals. 82 (5–6): 359–381. doi:10.1007/s11085-014-9496-1. OSTI 1185421. S2CID 136677636.
Bibliography
- Levitin, Valim (2006). High Temperature Strain of Metals and Alloys: Physical Fundamentals. WILEY-VCH. ISBN 978-3-527-31338-9.
- Shahsavari, H. A.; Kokabi, A. H.; Nategh, S. (2007). "Effect of preweld microstructure on HAZ liquation cracking of Rene 80 superalloy". Materials Science and Technology. 23 (5): 547–555. doi:10.1179/174328407x179539. S2CID 135755442.
External links
- "Superalloys". Cambridge University. Extensive bibliography and links.
