Cysteine dioxygenase
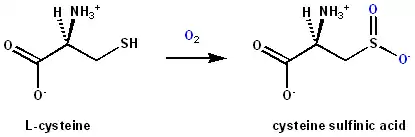
Cysteine dioxygenase (CDO) is a non-heme iron enzyme that catalyzes the conversion of L-cysteine to cysteine sulfinic acid (cysteine sulfinate). CDO plays an important role in cysteine catabolism, regulating intracellular levels of cysteine and responding changes in cysteine availability.[1] As such, CDO is highly regulated and undergoes large changes in concentration and efficiency. It oxidizes cysteine to the corresponding sulfinic acid by activation of dioxygen, although the exact mechanism of the reaction is still unclear. In addition to being found in mammals, CDO also exists in some yeast and bacteria, although the exact function is still unknown.[2][3] CDO has been implicated in various neurodegenerative diseases and cancers, which is likely related to cysteine toxicity.[1][2]
| Cysteine dioxygenase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 Human CDO (drawn from PDB 2IC1) | |||||||||
| Identifiers | |||||||||
| EC number | 1.13.11.20 | ||||||||
| CAS number | 37256-59-0 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| |||||||||
| cysteine dioxygenase, type I | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Symbol | CDO1 | ||||||
| NCBI gene | 1036 | ||||||
| HGNC | 1795 | ||||||
| OMIM | 603943 | ||||||
| RefSeq | NM_001801 | ||||||
| UniProt | Q16878 | ||||||
| Other data | |||||||
| EC number | 1.13.11.20 | ||||||
| Locus | Chr. 5 q23.2 | ||||||
| |||||||
Function
CDO is responsible for the first major step in metabolism of cysteine.[4] CDO oxidizes to cysteine sulfinic acid (which exists predominantly in the anionic sulfinate form in vivo). Overall, CDO catalyzes the addition of dioxygen (O2)[5] to a thiol, producing a sulfinic acid. More specifically, CDO is part of the group of non-heme iron oxygenases that employ oxygen as an electron acceptor. Cysteine sulfinic acid is then metabolized further via two divergent pathways: decarboxylated to hypotaurine by sulfinoalanine decarboxylase and oxidized to taurine by hypotaurine dehydrogenase; or transaminated to a putative 3-sulfinylpyruvate intermediate, which decomposes spontaneously into pyruvate and sulfite.[1][6] Sulfite can then be oxidized to sulfate by sulfite oxidase.[1] Thus CDO is necessary for hypotaurine/taurine and sulfite/sulfate production. The role of CDO may vary between cell types as it can either be used primarily for taurine or sulfate production or for degradation of cysteine.[1]
Structure
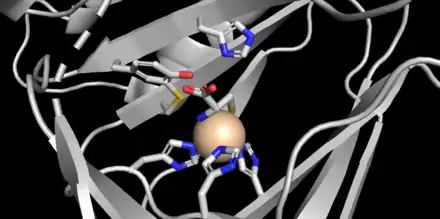
CDO is a 22.5 kDa protein[2] that contains 200 amino acid residues[3] and has an isoelectric point (pI) of 5.5.[2] The primary structure is highly conserved between mammalian species, with murine and human CDO differing in only 16 residues.[3] CDO is part of the cupin superfamily,[2] whose members possess a 6-stranded β-barrel[8] in a "jelly-roll" topology.[3] Crystal structures of the protein have been obtained at 1.5 Å resolution (mouse).[1] The active site displays a unique geometry where instead of the typical facial triad of two histidines and one carboxylate side-chain coordinating to an iron (II) species,[9] three histidine ligands are bound to iron.[2][3][8] Furthermore, crystal structures show the amino nitrogen and thiolate sulfur of cysteine coordinated to the iron in addition to a single water molecule (see figure).[2]
CDO contains a unique internal cofactor created by intramolecular thioether formation between Cys93 and Tyr157, which is postulated to participate in catalysis.[1] When the protein was first isolated, two bands on agarose gel were observed,[3] corresponding to the cofactor-containing protein and the unlinked "immature" protein, respectively. Crosslinking increases efficiency of CDO ten-fold and is regulated by levels of cysteine, an unusual example of protein cofactor formation mediated by substrate (feedforward activation).[1]
Mechanism
The CDO mechanism is still not well understood, despite active research to elucidate details of the reaction.[2] Overall, the reaction involves addition of O2 to cysteine, which occurs spontaneously without enzyme catalysis.[3] Studies have shown that the cysteinyltyrosine bridge lowers the oxidation potential of tyrosine (commonly an electron donor, as in photosystem II) by ~0.5 V relative to phenol and increases its acidity.[2] The thioether moiety likely plays a structural, redox, or, acid/base role. Other studies have shown that Tyr157 is needed for enzyme function (possibly as a tyrosinyl radical) and is highly conserved across CDO variants.[2] Furthermore, research has shown that cysteamine, a structurally similar molecule to cysteine, enhances cysteine oxidation but is not a substrate.[2][6]
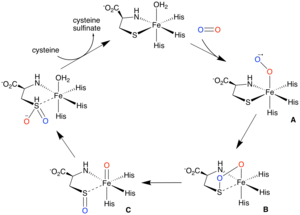
One proposed mechanism, supported by computational and spectroscopic studies, involves O2 binding cis to a thiolate to form reactive iron (III)-superoxo species (A), which then attacks the bound sulfur of cysteine to form a four-membered ring structure (B).[10][11][12] Heterolytic O-O bond cleavage then affords a high-valent iron (IV) oxo intermediate (C), which transfers the second oxygen to sulfur.[10][11]
Regulation
CDO is tightly regulated in the cell to maintain cysteine homeostasis. In particular, CDO responds to changes in dietary cysteine availability and protein intake, maintaining decreased activity with low cysteine levels and increased activity at high levels to prevent cytotoxicity.[1] Studies have shown that CDO can exhibit a dramatic increase in hepatic activity within hours. Unlike many enzymes, it is predominantly regulated at the level of protein turnover rather than transcriptional (mRNA levels). High cysteine levels inhibit ubiquitinylation, which lowers the rate of proteasomal degradation.[1] CDO is also regulated in adipose tissue, where high cysteine levels cause increased hypotaurine/taurine production.[1] Regulation of CDO is also thought to involve both the crosslinked and immature forms of the protein.
Disease Relevance
Because of its relevance to cysteine metabolism, changes in CDO activity may cause disease in humans. Research has found that elevated cysteine can by cytotoxic, neurotoxic,[1] and excitotoxic.[2] Abnormal or deficient CDO activity has been linked to Alzheimer's disease, Parkinson's disease, rheumatoid arthritis,[13] and motor neuron diseases.[1][2][14] In these diseases, patients display depressed sulfate levels, elevated fasting cysteine plasma concentrations, and other symptoms consistent with impaired cysteine oxidation.[1] CDO deficiency and subsequent cysteine accumulation in the globus pallidus has been linked to Hallervorden-Spatz disease.[15]
The expression of CDO is altered in cancer cells[2] and methylation of the CDO1 (human cysteine dioxygenase type I) promoter gene was shown to occur in colon, breast, esophageal, lung, bladder, and stomach cancers.[16] Silencing of CDO1 is a critical epigenetic event in breast cancer, leading to downregulation of CDO1 activity.[16][17] In particular, decreased CDO1 activity causes increased hydrogen sulfide (H2S), which has been connected to various diseases.[16] These results suggest that CDO1 (human cysteine dioxygenase type I) acts as a tumor suppressor gene and may potentially serve as a biomarker for cancer.[16]
References
- Stipanuk MH, Ueki I, Dominy JE, Simmons CR, Hirschberger LL (May 2009). "Cysteine dioxygenase: a robust system for regulation of cellular cysteine levels". Amino Acids. 37 (1): 55–63. doi:10.1007/s00726-008-0202-y. PMC 2736881. PMID 19011731.
- Joseph CA, Maroney MJ (August 2007). "Cysteine dioxygenase: structure and mechanism". Chemical Communications. 0 (32): 3338–49. doi:10.1039/B702158E. PMID 18019494.
- Stipanuk MH, Simmons CR, Karplus PA, Dominy JE (June 2011). "Thiol dioxygenases: unique families of cupin proteins". Amino Acids. 41 (1): 91–102. doi:10.1007/s00726-010-0518-2. PMC 3136866. PMID 20195658.
- Chai SC, Jerkins AA, Banik JJ, Shalev I, Pinkham JL, Uden PC, Maroney MJ (March 2005). "Heterologous expression, purification, and characterization of recombinant rat cysteine dioxygenase". The Journal of Biological Chemistry. 280 (11): 9865–9. doi:10.1074/jbc.M413733200. PMID 15623508.
- Lombardini JB, Singer TP, Boyer PD (March 1969). "Cystein oxygenase. II. Studies on the mechanism of the reaction with 18oxygen". The Journal of Biological Chemistry. 244 (5): 1172–5. PMID 5767301.
- Sakakibara S, Yamaguchi K, Hosokawa Y, Kohashi N, Ueda I (February 1976). "Purification and some properties of rat liver cysteine oxidase (cysteine dioxygenase)". Biochimica et Biophysica Acta (BBA) - Enzymology. 422 (2): 273–9. doi:10.1016/0005-2744(76)90138-8. PMID 2307.
- Ye S, Wu X, Wei L, Tang D, Sun P, Bartlam M, Rao Z (February 2007). "An insight into the mechanism of human cysteine dioxygenase. Key roles of the thioether-bonded tyrosine-cysteine cofactor". The Journal of Biological Chemistry. 282 (5): 3391–402. doi:10.1074/jbc.M609337200. PMID 17135237.
- McCoy JG, Bailey LJ, Bitto E, Bingman CA, Aceti DJ, Fox BG, Phillips GN (February 2006). "Structure and mechanism of mouse cysteine dioxygenase". Proceedings of the National Academy of Sciences of the United States of America. 103 (9): 3084–9. doi:10.1073/pnas.0509262103. PMC 1413891. PMID 16492780.
- Gardner JD, Pierce BS, Fox BG, Brunold TC (July 2010). "Spectroscopic and computational characterization of substrate-bound mouse cysteine dioxygenase: nature of the ferrous and ferric cysteine adducts and mechanistic implications". Biochemistry. 49 (29): 6033–41. doi:10.1021/bi100189h. PMC 2914100. PMID 20397631.
- Tchesnokov EP, Faponle AS, Davies CG, Quesne MG, Turner R, Fellner M, Souness RJ, Wilbanks SM, de Visser SP, Jameson GN (July 2016). "An iron-oxygen intermediate formed during the catalytic cycle of cysteine dioxygenase". Chemical Communications. 52 (57): 8814–7. doi:10.1039/C6CC03904A. PMC 5043143. PMID 27297454.
- Villar-Acevedo G, Lugo-Mas P, Blakely MN, Rees JA, Ganas AS, Hanada EM, Kaminsky W, Kovacs JA (January 2017). "Metal-Assisted Oxo Atom Addition to an Fe(III) Thiolate". Journal of the American Chemical Society. 139 (1): 119–129. doi:10.1021/jacs.6b03512. PMC 5262503. PMID 28033001.
- Kumar D, Thiel W, de Visser SP (March 2011). "Theoretical study on the mechanism of the oxygen activation process in cysteine dioxygenase enzymes". Journal of the American Chemical Society. 133 (11): 3869–82. doi:10.1021/ja107514f. PMID 21344861.
- Emery P, Bradley H, Arthur V, Tunn E, Waring R (July 1992). "Genetic factors influencing the outcome of early arthritis--the role of sulphoxidation status". British Journal of Rheumatology. 31 (7): 449–51. doi:10.1093/rheumatology/31.7.449. PMID 1628166.
- Heafield MT, Fearn S, Steventon GB, Waring RH, Williams AC, Sturman SG (March 1990). "Plasma cysteine and sulphate levels in patients with motor neurone, Parkinson's and Alzheimer's disease". Neuroscience Letters. 110 (1–2): 216–20. doi:10.1016/0304-3940(90)90814-p. PMID 2325885. S2CID 26672064.
- Perry TL, Norman MG, Yong VW, Whiting S, Crichton JU, Hansen S, Kish SJ (October 1985). "Hallervorden-Spatz disease: cysteine accumulation and cysteine dioxygenase deficiency in the globus pallidus". Annals of Neurology. 18 (4): 482–9. doi:10.1002/ana.410180411. PMID 4073841. S2CID 364798.
- Brait M, Ling S, Nagpal JK, Chang X, Park HL, Lee J, et al. (September 2012). "Cysteine dioxygenase 1 is a tumor suppressor gene silenced by promoter methylation in multiple human cancers". PLOS ONE. 7 (9): e44951. doi:10.1371/journal.pone.0044951. PMC 3459978. PMID 23028699.
- Jeschke J, O'Hagan HM, Zhang W, Vatapalli R, Calmon MF, Danilova L, et al. (June 2013). "Frequent inactivation of cysteine dioxygenase type 1 contributes to survival of breast cancer cells and resistance to anthracyclines". Clinical Cancer Research. 19 (12): 3201–11. doi:10.1158/1078-0432.CCR-12-3751. PMC 3985391. PMID 23630167.
External links
- CDO1 human gene location in the UCSC Genome Browser.
- CDO1 human gene details in the UCSC Genome Browser.