Pyrolysis
Pyrolysis is the thermal decomposition of materials at elevated temperatures in an inert atmosphere.[1] It involves a change of chemical composition. The word is coined from the Greek-derived elements pyro "fire" and lysis "separating".

Pyrolysis is most commonly used in the treatment of organic materials. It is one of the processes involved in charring wood.[2] In general, pyrolysis of organic substances produces volatile products and leaves a solid residue enriched in carbon, char. Extreme pyrolysis, which leaves mostly carbon as the residue, is called carbonization. Pyrolysis is considered as the first step in the processes of gasification or combustion.[3][4]
The process is used heavily in the chemical industry, for example, to produce ethylene, many forms of carbon, and other chemicals from petroleum, coal, and even wood, to produce coke from coal. Used also in the conversion of natural gas (primarily methane) into non-polluting hydrogen gas and non-polluting solid carbon char, initiating production in industrial volume.[5] Aspirational applications of pyrolysis would convert biomass into syngas and biochar, waste plastics back into usable oil, or waste into safely disposable substances.
Terminology
Pyrolysis is one of various types of chemical degradation processes that occur at higher temperatures (above the boiling point of water or other solvents). It differs from other processes like combustion and hydrolysis in that it usually does not involve the addition of other reagents such as oxygen (O2, in combustion) or water (in hydrolysis).[6] Pyrolysis produces solids (char), condensable liquids (tar), and uncondensing/permanent gasses.[7][8][9][10]
Types of pyrolysis
Complete pyrolysis of organic matter usually leaves a solid residue that consists mostly of elemental carbon; the process is then called carbonization. More specific cases of pyrolysis include:
- methane pyrolysis, in the presence of catalytic molten metals for the direct conversion of methane to non-polluting hydrogen fuel and separable solid carbon
- hydrous pyrolysis, in the presence of superheated water or steam, producing hydrogen and also substantial atmospheric carbon dioxide, also used in oil refining
- dry distillation, as in the original production of sulfuric acid from sulfates
- destructive distillation, as in the manufacture of charcoal, coke and activated carbon
- caramelization of sugars
- high-temperature cooking processes such as roasting, frying, toasting, and grilling
- charcoal burning, the production of charcoal
- tar production by pyrolysis of wood in tar kilns
- cracking of heavier hydrocarbons into lighter ones, as in oil refining
- thermal depolymerization, that breaks down plastics and other polymers into monomers and oligomers
- ceramization[11] involving the formation of polymer derived ceramics from preceramic polymers under an inert atmosphere
- catagenesis, the natural conversion of buried organic matter to fossil fuels and
- flash vacuum pyrolysis, used in organic synthesis.
General processes and mechanisms

Pyrolysis generally consists in heating the material above its decomposition temperature, breaking chemical bonds in its molecules. The fragments usually become smaller molecules, but may combine to produce residues with larger molecular mass, even amorphous covalent solids.
In many settings, some amounts of oxygen, water, or other substances may be present, so that combustion, hydrolysis, or other chemical processes may occur besides pyrolysis proper. Sometimes those chemical are added intentionally, as in the burning of firewood, in the traditional manufacture of charcoal, and in the steam cracking of crude oil.
Conversely, the starting material may be heated in a vacuum or in an inert atmosphere to avoid adverse chemical reactions. Pyrolysis in a vacuum also lowers the boiling point of the byproducts, improving their recovery.
When organic matter is heated at increasing temperatures in open containers, the following processes generally occur, in successive or overlapping stages:
- Below about 100 °C, volatiles, including some water, evaporate. Heat-sensitive substances, such as vitamin C and proteins, may partially change or decompose already at this stage.
- At about 100 °C or slightly higher, any remaining water that is merely absorbed in the material is driven off. Water trapped in crystal structure of hydrates may come off at somewhat higher temperatures. This process consumes a lot of energy, so the temperature may stop rising until this stage is complete.
- Some solid substances, like fats, waxes, and sugars, may melt and separate.
- Between 100 and 500 °C, many common organic molecules break down. Most sugars start decomposing at 160–180 °C. Cellulose, a major component of wood, paper, and cotton fabrics, decomposes at about 350 °C.[3] Lignin, another major wood component, starts decomposing at about 350 °C, but continues releasing volatile products up to 500 °C.[3] The decomposition products usually include water, carbon monoxide CO and/or carbon dioxide CO
2, as well as a large number of organic compounds.[4][12] Gases and volatile products leave the sample, and some of them may condense again as smoke. Generally, this process also absorbs energy. Some volatiles may ignite and burn, creating a visible flame. The non-volatile residues typically become richer in carbon and form large disordered molecules, with colors ranging between brown and black. At this point the matter is said to have been "charred" or "carbonized". - At 200–300 °C, if oxygen has not been excluded, the carbonaceous residue may start to burn, in a highly exothermic reaction, often with no or little visible flame. Once carbon combustion starts, the temperature rises spontaneously, turning the residue into a glowing ember and releasing carbon dioxide and/or monoxide. At this stage, some of the nitrogen still remaining in the residue may be oxidized into nitrogen oxides like NO
2 and N
2O
3. Sulfur and other elements like chlorine and arsenic may be oxidized and volatilized at this stage. - Once combustion of the carbonaceous residue is complete, a powdery or solid mineral residue (ash) is often left behind, consisting of inorganic oxidized materials of high melting point. Some of the ash may have left during combustion, entrained by the gases as fly ash or particulate emissions. Metals present in the original matter usually remain in the ash as oxides or carbonates, such as potash. Phosphorus, from materials such as bone, phospholipids, and nucleic acids, usually remains as phosphates.
Occurrence and uses
Cooking


Pyrolysis has many applications in food preparation.[13] Caramelization is the pyrolysis of sugars in food (often after the sugars have been produced by the breakdown of polysaccharides). The food goes brown and changes flavour. The distinctive flavours are used in many dishes; for instance, caramelized onion is used in French onion soup.[14][15] The temperatures needed for caramelization lie above the boiling point of water.[14] Frying oil can easily rise above boiling point. Putting a lid on the frying pan keeps the water in, and some of it re-condenses, keeping the temperature too cool to brown for longer.
Pyrolysis of food can also be undesirable, as in the charring of burnt food (at temperatures too low for the oxidative combustion of carbon to produce flames and burn the food to ash).
Coke, carbon, charcoals, and chars
Carbon and carbon-rich materials have desirable properties but are nonvolatile, even at high temperatures. Consequently, pyrolysis is used to produce many kinds of carbon; these can be used for fuel, as reagents in steelmaking (coke), and as structural materials.
Charcoal is a less smoky fuel than pyrolyzed wood).[16] Some cities ban, or used to ban, wood fires; when residents only use charcoal (and similarly-treated rock coal, called coke) air pollution is significantly reduced. In cities where people do not generally cook or heat with fires, this is not needed. In the mid-20th century, "smokeless" legislation in Europe required cleaner-burning techniques, such as coke fuel[17] and smoke-burning incinerators[18] as an effective measure to reduce air pollution[17]
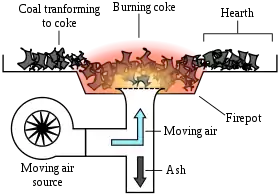
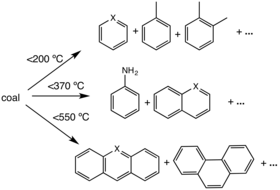
The coke-making or "coking" process consists of heating the material in "coking ovens" to very high temperatures (up to 900 °C or 1,700 °F) so that those molecules are broken down into lighter volatile substances, which leave the vessel, and a porous but hard residue that is mostly carbon and inorganic ash. The amount of volatiles varies with the source material, but is typically 25–30% of it by weight. High temperature pyrolysis is used on an industrial scale to convert coal into coke. This is useful in metallurgy, where the higher temperatures are necessary for many processes, such as steelmaking. Volatile by-products of this process are also often useful, including benzene and pyridine.[19] Coke can also be produced from the solid residue left from petroleum refining.
The original vascular structure of the wood and the pores created by escaping gases combine to produce a light and porous material. By starting with a dense wood-like material, such as nutshells or peach stones, one obtains a form of charcoal with particularly fine pores (and hence a much larger pore surface area), called activated carbon, which is used as an adsorbent for a wide range of chemical substances.
Biochar is the residue of incomplete organic pyrolysis, e.g., from cooking fires. They are a key component of the terra preta soils associated with ancient indigenous communities of the Amazon basin.[20] Terra preta is much sought by local farmers for its superior fertility and capacity to promote and retain an enhanced suite of beneficial microbiota, compared to the typical red soil of the region. Efforts are underway to recreate these soils through biochar, the solid residue of pyrolysis of various materials, mostly organic waste.

Carbon fibers are filaments of carbon that can be used to make very strong yarns and textiles. Carbon fiber items are often produced by spinning and weaving the desired item from fibers of a suitable polymer, and then pyrolyzing the material at a high temperature (from 1,500–3,000 °C or 2,730–5,430 °F). The first carbon fibers were made from rayon, but polyacrylonitrile has become the most common starting material. For their first workable electric lamps, Joseph Wilson Swan and Thomas Edison used carbon filaments made by pyrolysis of cotton yarns and bamboo splinters, respectively.
Pyrolysis is the reaction used to coat a preformed substrate with a layer of pyrolytic carbon. This is typically done in a fluidized bed reactor heated to 1,000–2,000 °C or 1,830–3,630 °F. Pyrolytic carbon coatings are used in many applications, including artificial heart valves.[21]
Liquid and gaseous biofuels
Pyrolysis is the basis of several methods for producing fuel from biomass, i.e. lignocellulosic biomass.[22] Crops studied as biomass feedstock for pyrolysis include native North American prairie grasses such as switchgrass and bred versions of other grasses such as Miscantheus giganteus. Other sources of organic matter as feedstock for pyrolysis include greenwaste, sawdust, waste wood, leaves, vegetables, nut shells, straw, cotton trash, rice hulls, and orange peels.[3] Animal waste including poultry litter, dairy manure, and potentially other manures are also under evaluation. Some industrial byproducts are also suitable feedstock including paper sludge, distillers grain,[23] and sewage sludge.[24]
In the biomass components, the pyrolysis of hemicellulose happens between 210 and 310 °C.[3] The pyrolysis of cellulose starts from 300-315 °C and ends at 360-380 °C, with a peak at 342-354 °C.[3] Lignin starts to decompose at about 200 °C and continues until 1000 °C.[25]
Synthetic diesel fuel by pyrolysis of organic materials is not yet economically competitive.[26] Higher efficiency is sometimes achieved by flash pyrolysis, in which finely divided feedstock is quickly heated to between 350 and 500 °C (660 and 930 °F) for less than two seconds.
Syngas is usually produced by pyrolysis.[13]
The low quality of oils produced through pyrolysis can be improved by physical and chemical processes,[27] which might drive up production costs, but may make sense economically as circumstances change.
There is also the possibility of integrating with other processes such as mechanical biological treatment and anaerobic digestion.[28] Fast pyrolysis is also investigated for biomass conversions.[29] Fuel bio-oil can also be produced by hydrous pyrolysis.
Methane pyrolysis for Hydrogen
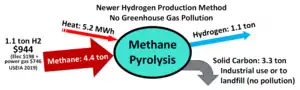
Methane pyrolysis[30] is a non-polluting industrial process for hydrogen production from methane by removing solid carbon from natural gas. This one step process produces non-polluting hydrogen in high volume at low cost. Only water is released when hydrogen is used as the fuel for fuel-cell electric heavy truck transportation,[31][32][33][34][35] gas turbine electric power generation,[36][37] and hydrogen for industrial processes.[38] Methane pyrolysis is the process operating around 1065 °C for producing hydrogen from natural gas that allows removal of carbon easily (solid non-polluting carbon is a byproduct of the process).[39][40] The industrial quality carbon can then be sold or landfilled and is not released into the atmosphere, no emission of greenhouse gas (GHG). Volume production is being evaluated in the BASF "methane pyrolysis at scale" pilot plant,[41] in such research laboratories as Karlsruhe Liquid-metal Laboratory (KALLA)[42] and the chemical engineering team at University of California - Santa Barbara.[43] Electricity consumption is only one seventh of the amount consumed in the water electrolysis method for producing hydrogen.[44]
Ethylene
Pyrolysis is used to produce ethylene, the chemical compound produced on the largest scale industrially (>110 million tons/year in 2005). In this process, hydrocarbons from petroleum are heated to around 600 °C (1,112 °F) in the presence of steam; this is called steam cracking. The resulting ethylene is used to make antifreeze (ethylene glycol), PVC (via vinyl chloride), and many other polymers, such as polyethylene and polystyrene.[45]
Semiconductors
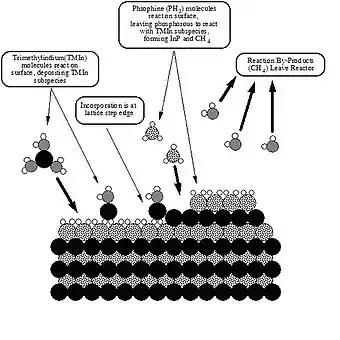
The process of metalorganic vapour phase epitaxy (MOCVD) entails pyrolysis of volatile organometallic compounds to give semiconductors, hard coatings, and other applicable materials. The reactions entail thermal degradation of precursors, with deposition of the inorganic component and release of the hydrocarbons as gaseous waste. Since it is an atom-by-atom deposition, these atoms organize themselves into crystals to form the bulk semiconductor. Silicon chips are produced by the pyrolysis of silane:
- SiH4 → Si + 2 H2.
Gallium arsenide, another semiconductor, forms upon co-pyrolysis of trimethylgallium and arsine.
Waste management
Pyrolysis can also be used to treat municipal solid waste and plastic waste.[4][12][46] The main advantage is the reduction in volume of the waste. In principle, pyrolysis will regenerate the monomers (precursors) to the polymers that are treated, but in practice the process is neither a clean nor an economically competitive source of monomers.[47][48][49]
In tire waste management, tire pyrolysis is well developed technology.[50] Other products from car tire pyrolysis include steel wires, carbon black and bitumen.[51] The area faces legislative, economic, and marketing obstacles.[52] Oil derived from tire rubber pyrolysis contains high sulfur content, which gives it high potential as a pollutant and should be desulfurized.[53][54]
Alkaline pyrolysis of sewage sludge at low temperature of 500 °C can enhance H2 production with in-situ carbon capture. The use of NaOH as has the potential to produce H2-rich gas that can be used for fuels cells directly.[24][55]
Thermal cleaning
Pyrolysis is also used for thermal cleaning, an industrial application to remove organic substances such as polymers, plastics and coatings from parts, products or production components like extruder screws, spinnerets[56] and static mixers. During the thermal cleaning process, at temperatures between 310 C° to 540 C° (600 °F to 1000 °F),[57] organic material is converted by pyrolysis and oxidation into volatile organic compounds, hydrocarbons and carbonized gas.[58] Inorganic elements remain.[59]
Several types of thermal cleaning systems use pyrolysis:
- Molten Salt Baths belong to the oldest thermal cleaning systems; cleaning with a molten salt bath is very fast but implies the risk of dangerous splatters, or other potential hazards connected with the use of salt baths, like explosions or highly toxic hydrogen cyanide gas.[57]
- Fluidized Bed Systems[60] use sand or aluminium oxide as heating medium;[61] these systems also clean very fast but the medium does not melt or boil, nor emit any vapors or odors;[57] the cleaning process takes one to two hours.[58]
- Vacuum Ovens use pyrolysis in a vacuum[62] avoiding uncontrolled combustion inside the cleaning chamber;[57] the cleaning process takes 8[58] to 30 hours.[63]
- Burn-Off Ovens, also known as Heat-Cleaning Ovens, are gas-fired and used in the painting, coatings, electric motors and plastics industries for removing organics from heavy and large metal parts.[64]
Fine chemical synthesis
Pyrolysis is used in the production of chemical compounds, mainly, but not only, in the research laboratory.
The area of boron-hydride clusters started with the study of the pyrolysis of diborane (B2H6) at ca. 200 °C. Products include the clusters pentaborane and decaborane. These pyrolyses involve not only cracking (to give H2), but also recondensation.[65]
The synthesis of nanoparticles,[66] zirconia[67] and oxides[68] utilizing an ultrasonic nozzle in a process called ultrasonic spray pyrolysis (USP).
Other uses and occurrences
- Pyrolysis is used to turn organic materials into carbon for the purpose of carbon-14 dating.
- Pyrolysis of tobacco, paper, and additives, in cigarettes and other products, generates many volatile products (including nicotine, carbon monoxide, and tar) that are responsible for the aroma and health effects of smoking. Similar considerations apply to the smoking of marijuana and the burning of incense products and mosquito coils.
- Pyrolysis occurs during the incineration of trash, potentially generating volatiles that are toxic or contribute to air pollution if not completely burned.
- Laboratory or industrial equipment sometimes gets fouled by carbonaceous residues that result from coking, the pyrolysis of organic products that come into contact with hot surfaces.
PAHs generation
Polycyclic aromatic hydrocarbons (PAHs) can be generated from the pyrolysis of different solid waste fractions,[10] such as hemicellulose, cellulose, lignin, pectin, starch, polyethylene (PE), polystyrene (PS), polyvinyl chloride (PVC), and polyethylene terephthalate (PET). PS, PVC, and lignin generate significant amount of PAHs. Naphthalene is the most abundant PAH among all the polycyclic aromatic hydrocarbons.[69]
When the temperature is increased from 500 to 900 °C, most PAHs increase. With the increase of the temperature, the percentage of light PAHs decrease and the percentage of heavy PAHs increase.[70][71]
Study tools
Thermogravimetric analysis
Thermogravimetric analysis (TGA) is one of the most common techniques to investigate pyrolysis with no limitations of heat and mass transfer. The results can be used to determine mass loss kinetics.[3][12][4][25][46] Activation energies can be calculated using Kissinger method or peak analysis-least square method (PA-LSM).[4][25]
TGA can couple with Fourier-transform infrared spectroscopy (FTIR) and mass spectrometry. As the increase of temperature, the volatiles generated from pyrolysis can be measured.[72][55]
Macro-TGA
In TGA, sample is loaded first before the increase of temperature, and the heating rate is low (less than 100 °C min−1). Macro-TGA can use gram level samples which can be used to investigate the pyrolysis with mass and heat transfer effects.[4][73]
Pyrolysis–gas chromatography–mass spectrometry
Pyrolysis mass spectrometry (Py-GC-MS) is an important laboratory procedure to determine the structure of compounds.[74][75]
History

Pyrolysis has been used for turning wood into charcoal since ancient times. In their embalming process, the ancient Egyptians used methanol, which they obtained from the pyrolysis of wood. The dry distillation of wood remained the major source of methanol into the early 20th century.[76]
Pyrolysis was instrumental in the discovery of many important chemical substances, such as phosphorus (from ammonium sodium hydrogen phosphate NH
4NaHPO
4 in concentrated urine) and oxygen (from mercuric oxide and various nitrates).
See also
References
- "Pyrolysis". Compendium of Chemical Terminology. International Union of Pure and Applied Chemistry. 2009. p. 1824. doi:10.1351/goldbook.P04961. ISBN 978-0-9678550-9-7. Retrieved 2018-01-10.
- Burning of wood Archived 2010-02-09 at the Wayback Machine, InnoFireWood's website. Accessed on 2010-02-06.
- Zhou, Hui; Long, YanQiu; Meng, AiHong; Li, QingHai; Zhang, YanGuo (August 2013). "The pyrolysis simulation of five biomass species by hemi-cellulose, cellulose and lignin based on thermogravimetric curves". Thermochimica Acta. 566: 36–43. doi:10.1016/j.tca.2013.04.040.
- Zhou, Hui (2017). "Combustible Solid Waste Thermochemical Conversion". Springer Theses. doi:10.1007/978-981-10-3827-3. ISBN 978-981-10-3826-6. ISSN 2190-5053. S2CID 135947379.
- BASF. "BASF researchers working on fundamentally new, low-carbon production processes, Methane Pyrolysis". United States Sustainability. BASF. Retrieved 19 October 2020.
- Cory A. Kramer, Reza Loloee, Indrek S. Wichman and Ruby N. Ghosh, 2009, Time Resolved Measurements of Pyrolysis Products From Thermoplastic Poly-Methyl-Methacrylate (PMMA) Archived 2014-11-06 at the Wayback Machine ASME 2009 International Mechanical Engineering Congress and Exposition
- Ramin, L.; Assadi, M. Hussein N.; Sahajwalla, V. (2014). "High-density polyethylene degradation into low molecular weight gases at 1823K: An atomistic simulation". J. Anal. Appl. Pyrol. 110: 318–321. doi:10.1016/j.jaap.2014.09.022.
- Jones, Jim. "Mechanisms of pyrolysis" (PDF). Retrieved 19 May 2019.
- George, Anthe; Turn, Scott Q.; Morgan, Trevor James (26 August 2015). "Fast Pyrolysis Behavior of Banagrass as a Function of Temperature and Volatiles Residence Time in a Fluidized Bed Reactor". PLOS ONE. 10 (8): e0136511. Bibcode:2015PLoSO..1036511M. doi:10.1371/journal.pone.0136511. ISSN 1932-6203. PMC 4550300. PMID 26308860.
- Zhou, Hui; Wu, Chunfei; Meng, Aihong; Zhang, Yanguo; Williams, Paul T. (November 2014). "Effect of interactions of biomass constituents on polycyclic aromatic hydrocarbons (PAH) formation during fast pyrolysis" (PDF). Journal of Analytical and Applied Pyrolysis. 110: 264–269. doi:10.1016/j.jaap.2014.09.007.
- Wang, Xifan; Schmidt, Franziska; Hanaor, Dorian; Kamm, Paul H.; Li, Shuang; Gurlo, Aleksander (2019). "Additive manufacturing of ceramics from preceramic polymers: A versatile stereolithographic approach assisted by thiol-ene click chemistry". Additive Manufacturing. 27: 80–90. arXiv:1905.02060. Bibcode:2019arXiv190502060W. doi:10.1016/j.addma.2019.02.012. S2CID 104470679.
- Zhou, Hui; Long, YanQiu; Meng, AiHong; Li, QingHai; Zhang, YanGuo (April 2015). "Thermogravimetric characteristics of typical municipal solid waste fractions during co-pyrolysis". Waste Management. 38: 194–200. doi:10.1016/j.wasman.2014.09.027. PMID 25680236.
- Kaplan, Ryan (Fall 2011). "Pyrolysis: Biochar, Bio-Oil and Syngas from Wastes" (Course notes for Environmental Resources Engineering 115). users.humboldt.edu. Humboldt University. Retrieved 19 May 2019.
- "What is Caramelization?". www.scienceofcooking.com. Retrieved 19 May 2019.
- Brimm, Courtney (7 November 2011). "Cooking with Chemistry: What is Caramelization?". Common Sense Science. Retrieved 19 May 2019.
- Sood, A (December 2012). "Indoor fuel exposure and the lung in both developing and developed countries: an update". Clinics in Chest Medicine. 33 (4): 649–65. doi:10.1016/j.ccm.2012.08.003. PMC 3500516. PMID 23153607.
- "SMOKELESS zones". British Medical Journal. 2 (4840): 818–20. 10 October 1953. doi:10.1136/bmj.2.4840.818. PMC 2029724. PMID 13082128.
- Smokeless incinerator patent
- Ludwig Briesemeister, Andreas Geißler, Stefan Halama, Stephan Herrmann, Ulrich Kleinhans, Markus Steibel, Markus Ulbrich, Alan W. Scaroni, M. Rashid Khan, Semih Eser, Ljubisa R. Radovic (2002). "Coal Pyrolysis". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. pp. 1–44. doi:10.1002/14356007.a07_245.pub2. ISBN 9783527306732.CS1 maint: uses authors parameter (link)
- Lehmann, Johannes. "Biochar: the new frontier". Archived from the original on 2008-06-18. Retrieved 2008-07-10.
- Ratner, Buddy D. (2004). Pyrolytic carbon. In Biomaterials science: an introduction to materials in medicine Archived 2014-06-26 at the Wayback Machine. Academic Press. pp. 171–180. ISBN 0-12-582463-7.
- Evans, G. "Liquid Transport Biofuels – Technology Status Report" Archived September 19, 2008, at the Wayback Machine, "National Non-Food Crops Centre", 14-04-08. Retrieved on 2009-05-05.
- "Biomass Feedstock for Slow Pyrolysis". BEST Pyrolysis, Inc. website. BEST Energies, Inc. Archived from the original on 2012-01-02. Retrieved 2010-07-30.
- Zhao, Ming; Wang, Fan; Fan, Yiran; Raheem, Abdul; Zhou, Hui (March 2019). "Low-temperature alkaline pyrolysis of sewage sludge for enhanced H2 production with in-situ carbon capture". International Journal of Hydrogen Energy. 44 (16): 8020–8027. doi:10.1016/j.ijhydene.2019.02.040.
- Zhou, Hui; Long, Yanqiu; Meng, Aihong; Chen, Shen; Li, Qinghai; Zhang, Yanguo (2015). "A novel method for kinetics analysis of pyrolysis of hemicellulose, cellulose, and lignin in TGA and macro-TGA". RSC Advances. 5 (34): 26509–26516. doi:10.1039/C5RA02715B. ISSN 2046-2069.
- "Pyrolysis and Other Thermal Processing". US DOE. Archived from the original on 2007-08-14.
- Ramirez, Jerome; Brown, Richard; Rainey, Thomas (1 July 2015). "A Review of Hydrothermal Liquefaction Bio-Crude Properties and Prospects for Upgrading to Transportation Fuels". Energies. 8 (7): 6765–6794. doi:10.3390/en8076765.
- Marshall, A. T. & Morris, J. M. (2006) A Watery Solution and Sustainable Energy Parks Archived 2007-09-28 at the Wayback Machine, CIWM Journal, pp. 22–23
- Westerhof, Roel Johannes Maria (2011). Refining fast pyrolysis of biomass. Thermo-Chemical Conversion of Biomass (Thesis). University of Twente. Archived from the original on 2013-06-17. Retrieved 2012-05-30.
- Upham, D. Chester. "Catalytic molten metals for the direct conversion of methane to hydrogen and separable carbon in a single reaction step commercial process (at potentially low-cost). This would provide no-pollution hydrogen from natural gas, essentially forever". ScienceMag.org. American Association for Advancement of Science. Retrieved 31 October 2020.
- Fialka, John. "Energy Department Looks to Boost Hydrogen Fuel for Big Trucks". E&E News. Scientific American. Retrieved 7 November 2020.
- CCJ News. "How fuel cell trucks produce electric power and how they're fueled". CCJ News. Commercial Carrier Journal. Retrieved 19 October 2020.
- Toyota. "Hydrogen Fuel-Cell Class 8 Truck". Hydrogen-Powered Truck Will Offer Heavy-Duty Capability and Clean Emissions. Toyota. Retrieved 19 October 2020.
-
Colias, Mike. [Auto Makers Shift Hydrogen Focus to Big Rigs https://www.wsj.com/articles/auto-makers-shift-their-hydrogen-focus-to-big-rigs-11603714573 "Auto Makers Shift Their Hydrogen Focus to Big Rigs"] Check
|url=value (help). Wall Street Journal. Retrieved 26 October 2020. - Honda. "Honda Fuel-Cell Clarity". Clarity Fuel Cell. Honda. Retrieved 19 October 2020.
- GE Turbines. "Hydrogen fueled power turbines". Hydrogen fueled gas turbines. General Electric. Retrieved 19 October 2020.
- Solar Turbines. "Hydrogen fueled power turbines". Power From Hydrogen Gas For Carbon Reduction. Solar Turbines. Retrieved 19 October 2020.
- Crolius, Stephen H. "Methane to Ammonia via Pyrolysis". Ammonia Energy Association. Ammonia Energy Association. Retrieved 19 October 2020.
- Cartwright, Jon. "The reaction that would give us clean fossil fuels forever". NewScientist. New Scientist Ltd. Retrieved 30 October 2020.
- Karlsruhe Institute of Technology. "Hydrogen from methane without CO2 emissions". Phys.Org. Phys.Org. Retrieved 30 October 2020.
- BASF. "BASF researchers working on fundamentally new, low-carbon production processes, Methane Pyrolysis". United States Sustainability. BASF. Retrieved 19 October 2020.
- Gusev, Alexander. "KITT/IASS - Producing CO2 Free Hydrogen From Natural Gas For Energy Usage". European Energy Innovation. Institute for Advanced Sustainability Studies. Retrieved 30 October 2020.
- Fernandez, Sonia. "Journalist". Phys-Org. American Institute of Physics. Retrieved 19 October 2020.
- "Methane pyrolysis process uses renewable electricity split CH4 into H2 and carbon-black". Retrieved 17 December 2020.
- Zimmermann, Heinz; Walz, Roland (2008). "Ethylene". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a10_045.pub3. ISBN 978-3527306732.
- Zhou, Hui; Long, YanQiu; Meng, AiHong; Li, QingHai; Zhang, YanGuo (January 2015). "Interactions of three municipal solid waste components during co-pyrolysis". Journal of Analytical and Applied Pyrolysis. 111: 265–271. doi:10.1016/j.jaap.2014.08.017.
- Kaminsky, Walter (2000). "Plastics, Recycling". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a21_057. ISBN 978-3527306732.
- N.J. Themelis et al. "Energy and Economic Value of Nonrecyclable Plastics and Municipal Solid Wastes that are Currently Landfilled in the Fifty States" Columbia University Earth Engineering Center Archived 2014-05-08 at the Wayback Machine
- The Plastic to Oil Machine | A\J – Canada's Environmental Voice Archived 2015-09-09 at the Wayback Machine. Alternativesjournal.ca (2016-12-07). Retrieved on 2016-12-16.
- ผศ.ดร.ศิริรัตน์ จิตการค้า, "ไพโรไลซิสยางรถยนต์หมดสภาพ : กลไกการผลิตน้ำมันเชื้อเพลิงคุณภาพสูง"วิทยาลัยปิโตรเลียมและปิโตรเคมี จุฬาลงกรณ์มหาวิทยาลัย (in Thai) Jidgarnka, S. "Pyrolysis of Expired Car Tires: Mechanics of Producing High Quality Fuels" Archived 2015-02-20 at the Wayback Machine. Chulalongkorn University Department of Petrochemistry
- Roy, C.; Chaala, A.; Darmstadt, H. (1999). "The vacuum pyrolysis of used tires". Journal of Analytical and Applied Pyrolysis. 51 (1–2): 201–221. doi:10.1016/S0165-2370(99)00017-0.
- Martínez, Juan Daniel; Puy, Neus; Murillo, Ramón; García, Tomás; Navarro, María Victoria; Mastral, Ana Maria (2013). "Waste tyre pyrolysis – A review, Renewable and Sustainable". Energy Reviews. 23: 179–213. doi:10.1016/j.rser.2013.02.038.
- Choi, G.-G.; Jung, S.-H.; Oh, S.-J.; Kim, J.-S. (2014). "Total utilization of waste tire rubber through pyrolysis to obtain oils and CO2 activation of pyrolysis char". Fuel Processing Technology. 123: 57–64. doi:10.1016/j.fuproc.2014.02.007.
- Ringer, M.; Putsche, V.; Scahill, J. (2006) Large-Scale Pyrolysis Oil Production: A Technology Assessment and Economic Analysis Archived 2016-12-30 at the Wayback Machine; NREL/TP-510-37779; National Renewable Energy Laboratory (NREL), Golden, CO.
- Zhao, Ming; Memon, Muhammad Zaki; Ji, Guozhao; Yang, Xiaoxiao; Vuppaladadiyam, Arun K.; Song, Yinqiang; Raheem, Abdul; Li, Jinhui; Wang, Wei; Zhou, Hui (April 2020). "Alkali metal bifunctional catalyst-sorbents enabled biomass pyrolysis for enhanced hydrogen production". Renewable Energy. 148: 168–175. doi:10.1016/j.renene.2019.12.006.
- Heffungs, Udo (June 2010). "Effective Spinneret Cleaning". Fiber Journal. Archived from the original on 30 June 2016. Retrieved 19 April 2016.
- Mainord, Kenneth (September 1994). "Cleaning with Heat: Old Technology with a Bright New Future" (PDF). Pollution Prevention Regional Information Center. The Magazine of Critical Cleaning Technology. Archived (PDF) from the original on 8 December 2015. Retrieved 4 December 2015.
- "A Look at Thermal Cleaning Technology". ThermalProcessing.org. Process Examiner. 14 March 2014. Archived from the original on 8 December 2015. Retrieved 4 December 2015.
- Davis, Gary; Brown, Keith (April 1996). "Cleaning Metal Parts and Tooling" (PDF). Pollution Prevention Regional Information Center. Process Heating. Archived (PDF) from the original on 4 March 2016. Retrieved 4 December 2015.
- Schwing, Ewald; Uhrner, Horst (7 October 1999). "Method for removing polymer deposits which have formed on metal or ceramic machine parts, equipment and tools". Espacenet. European Patent Office. Retrieved 19 April 2016.
- Staffin, Herbert Kenneth; Koelzer, Robert A. (28 November 1974). "Cleaning objects in hot fluidised bed – with neutralisation of resultant acidic gas esp. by alkaline metals cpds". Espacenet. European Patent Office. Retrieved 19 April 2016.
- Dwan, Thomas S. (2 September 1980). "Process for vacuum pyrolysis removal of polymers from various objects". Espacenet. European Patent Office. Retrieved 26 December 2015.
- "Vacuum pyrolysis systems". thermal-cleaning.com. Archived from the original on 15 February 2016. Retrieved 11 February 2016.
- "Paint Stripping: Reducing Waste and Hazardous Material". Minnesota Technical Assistance Program. University of Minnesota. July 2008. Archived from the original on 8 December 2015. Retrieved 4 December 2015.
- Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8. gives Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 0-08-037941-9.
- Pingali, Kalyana C.; Rockstraw, David A.; Deng, Shuguang (2005). "Silver Nanoparticles from Ultrasonic Spray Pyrolysis of Aqueous Silver Nitrate" (PDF). Aerosol Science and Technology. 39 (10): 1010–1014. Bibcode:2005AerST..39.1010P. doi:10.1080/02786820500380255. S2CID 6908181. Archived (PDF) from the original on 2014-04-08.
- Song, Y. L.; Tsai, S. C.; Chen, C. Y.; Tseng, T. K.; Tsai, C. S.; Chen, J. W.; Yao, Y. D. (2004). "Ultrasonic Spray Pyrolysis for Synthesis of Spherical Zirconia Particles" (PDF). Journal of the American Ceramic Society. 87 (10): 1864–1871. doi:10.1111/j.1151-2916.2004.tb06332.x. Archived (PDF) from the original on 2014-04-08.
- Hamedani, Hoda Amani (2008) Investigation of Deposition Parameters in Ultrasonic Spray Pyrolysis for Fabrication of Solid Oxide Fuel Cell Cathode Archived 2016-03-05 at the Wayback Machine, Georgia Institute of Technology
- Zhou, Hui; Wu, Chunfei; Onwudili, Jude A.; Meng, Aihong; Zhang, Yanguo; Williams, Paul T. (February 2015). "Polycyclic aromatic hydrocarbons (PAH) formation from the pyrolysis of different municipal solid waste fractions" (PDF). Waste Management. 36: 136–146. doi:10.1016/j.wasman.2014.09.014. PMID 25312776.
- Zhou, Hui; Wu, Chunfei; Onwudili, Jude A.; Meng, Aihong; Zhang, Yanguo; Williams, Paul T. (2014-10-16). "Polycyclic Aromatic Hydrocarbon Formation from the Pyrolysis/Gasification of Lignin at Different Reaction Conditions". Energy & Fuels. 28 (10): 6371–6379. doi:10.1021/ef5013769. ISSN 0887-0624.
- Zhou, Hui; Wu, Chunfei; Onwudili, Jude A.; Meng, Aihong; Zhang, Yanguo; Williams, Paul T. (April 2016). "Influence of process conditions on the formation of 2–4 ring polycyclic aromatic hydrocarbons from the pyrolysis of polyvinyl chloride" (PDF). Fuel Processing Technology. 144: 299–304. doi:10.1016/j.fuproc.2016.01.013.
- Zhou, Hui; Meng, AiHong; Long, YanQiu; Li, QingHai; Zhang, YanGuo (July 2014). "Interactions of municipal solid waste components during pyrolysis: A TG-FTIR study". Journal of Analytical and Applied Pyrolysis. 108: 19–25. doi:10.1016/j.jaap.2014.05.024.
- Long, Yanqiu; Zhou, Hui; Meng, Aihong; Li, Qinghai; Zhang, Yanguo (September 2016). "Interactions among biomass components during co-pyrolysis in (macro)thermogravimetric analyzers". Korean Journal of Chemical Engineering. 33 (9): 2638–2643. doi:10.1007/s11814-016-0102-x. ISSN 0256-1115. S2CID 59127489.
- Goodacre, R.; Kell, D. B. (1996). "Pyrolysis mass spectrometry and its applications in biotechnology". Curr. Opin. Biotechnol. 7 (1): 20–28. doi:10.1016/S0958-1669(96)80090-5. PMID 8791308.CS1 maint: uses authors parameter (link)
- Peacock, P. M.; McEwen, C. N. (2006). "Mass Spectrometry of Synthetic Polymers. Anal. Chem". Analytical Chemistry. 78 (12): 3957–3964. doi:10.1021/ac0606249. PMID 16771534.CS1 maint: uses authors parameter (link)
- E. Fiedler, G. Grossmann, D. B. Kersebohm, G. Weiss, Claus Witte (2005). "Methanol". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007. ISBN 978-3527306732.CS1 maint: uses authors parameter (link)
