Orthornavirae
Orthornavirae is a kingdom of viruses that have genomes made of ribonucleic acid (RNA) and which encode an RNA-dependent RNA polymerase (RdRp). The RdRp is used to transcribe the viral RNA genome into messenger RNA (mRNA) and to replicate the genome. Viruses in this kingdom also share a number of characteristics involving evolution, including high rates of genetic mutations, recombinations, and reassortments.
| Orthornavirae | |
|---|---|
| Virus classification | |
| (unranked): | Virus |
| Realm: | Riboviria |
| Kingdom: | Orthornavirae |
| Phyla and classes | |
Viruses in Orthornavirae belong to the realm Riboviria. They are descended from a common ancestor that may have been a non-viral molecule that encoded a reverse transcriptase instead of an RdRp for replication. The kingdom is subdivided into five phyla that separate member viruses based on their genome type, host range, and genetic similarity. Viruses with three genome types are included: positive-strand RNA viruses, negative-strand RNA viruses, and double-stranded RNA viruses.
Many of the most widely known viral diseases are caused by RNA viruses in the kingdom, which includes coronaviruses, the Ebola virus, influenza viruses, the measles virus, and the rabies virus. The first virus to be discovered, tobacco mosaic virus, belongs to the kingdom. In modern history, RdRp-encoding RNA viruses have caused numerous disease outbreaks, and they infect many economically important crops. Most eukaryotic viruses, including most human, animal, and plant viruses, are RdRp-encoding RNA viruses. In contrast, there are relatively few prokaryotic viruses in the kingdom.
Etymology
The first part of Orthornavirae comes from Greek ὀρθός [orthós], meaning straight, the middle part, rna, refers to RNA, and -virae is the suffix used for virus kingdoms.[1]
Characteristics
Structure

RNA viruses in Orthornavirae typically do not encode that many proteins, but most positive-sense, single-stranded (+ssRNA) viruses and some double-stranded RNA (dsRNA) viruses encode a major capsid protein that has a single jelly roll fold, so named because the folded structure of the protein contains a structure that resembles a jelly roll.[2] Many also encode an envelope, a type of lipid membrane that typically surrounds the capsid. In particular, the viral envelope is near-universal among negative-sense, single-stranded (-ssRNA) viruses.[3][4]
Genome
Viruses in Orthornavirae have three different types of genomes: dsRNA, +ssRNA, and -ssRNA. Single-stranded RNA viruses have either a positive or negative sense strand, and dsRNA viruses have both. This structure of the genome is important in terms of transcription to synthesize viral mRNA as well as replication of the genome, both of which are carried out by the viral enzyme RNA-dependent RNA polymerase (RdRp), also called RNA replicase.[1][2]
Positive-strand RNA viruses
Positive-strand RNA viruses have genomes that can function as mRNA, so transcription is not necessary. However, +ssRNA will produce dsRNA forms as part of the process of replicating their genomes. From the dsRNA, additional positive strands are synthesized, which may be used as mRNA or for genomes for progeny. Because +ssRNA viruses create intermediate dsRNA forms, they have to avoid the host's immune system in order to replicate. +ssRNA viruses accomplish this by replicating in membrane-associated vesicles that are used as replication factories. For many +ssRNA viruses, subgenomic portions of the genome will be transcribed to translate specific proteins, whereas others will transcribe a polyprotein that is cleaved to produce separate proteins.[5][6]
Negative-strand RNA viruses
Negative-strand RNA viruses have genomes that function as templates from which mRNA can be synthesized directly by RdRp.[7] Replication is the same process but executed on the positive sense antigenome, during which RdRp ignores all transcription signals so that a complete -ssRNA genome can be synthesized.[8] -ssRNA viruses vary between those that inititate transcription by the RdRp creating a cap on the 5'-end (usually pronounced "five prime end") of the genome or by snatching a cap from host mRNA and attaching it to the viral RNA.[9] For many -ssRNA viruses, at the end of transcription, RdRp stutters on a uracil in the genome, synthesizing hundreds of adenines in a row as part of creating a polyadenylated tail for the mRNA.[10] Some -ssRNA viruses are essentially ambisense, and have proteins encoded by both the positive and negative strand, so mRNA is synthesized directly from the genome and from a complementary strand.[11]
Double-stranded RNA viruses
For dsRNA viruses, RdRp transcribes mRNA by using the negative strand as a template. Positive strands may also be used as templates to synthesize negative strands for the construction of genomic dsRNA. dsRNA is not a molecule produced by cells, so cellular life has evolved mechanisms to detect and inactivate viral dsRNA. To counter this, dsRNA viruses typically retain their genomes inside of viral capsid in order to evade the host's immune system.[12]
Evolution
RNA viruses in Orthornavirae experience a high rate of genetic mutations because RdRp is prone to making errors in replication since it typically lacks proofreading mechanisms to repair errors.[note 1] Mutations in RNA viruses are often influenced by host factors such as dsRNA-dependent adenosine deaminases, which edit viral genomes by changing adenosines to inosines.[13][14] Mutations in genes that are essential for replication lead to a reduced number of progeny, so viral genomes typically contain sequences that are highly conserved over time with relatively few mutations.[15]
Many RdRp-encoding RNA viruses also experience a high rate of genetic recombination, though rates of recombination vary significantly, with lower rates in -ssRNA viruses and higher rates in dsRNA and +ssRNA viruses. There are two types of recombination: copy choice recombination and ressortment. Copy choice recombination occurs when the RdRp switches templates during synthesis without releasing the prior, newly created RNA strand, which generates a genome of mixed ancestry. Reassortment, which is restricted to viruses with segmented genomes, has segments from different genomes packaged into a single virion, or virus particle, which also produces hybrid progeny.[13][16]
For reassortment, some segmented viruses package their genomes into multiple virions, which produces genomes that are random mixtures of parents, whereas for those that are packaged into a single virion, typically individual segments are swapped. Both forms of recombination can only occur if more than one virus is present in a cell, and the more alleles are present, the more likely recombination is to occur. A key difference between copy choice recombination and reassortment is that copy choice recombination can occur anywhere in a genome, whereas reassortment swaps fully-replicated segments. Therefore, copy choice recombination can produce non-functional viral proteins whereas reassortment cannot.[13][16][17][18]
The mutation rate of a virus is associated with the rate of genetic recombinations. Higher mutation rates increase both the number of advantageous and disadvantageous mutations, whereas higher rates of recombination allows for beneficial mutations to be separated from deleterious ones. Therefore, higher rates of mutations and recombinations, up to a certain point, improve viruses' ability to adapt.[13][19] Notable examples of this include reassortments that enable cross-species transmission of influenza viruses, which have led to numerous pandemics, as well as the emergence of drug-resistance influenza strains via mutations that were reassorted.[18]
Phylogenetics
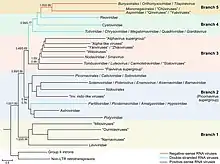
The exact origin of Orthornavirae is not well established, but the viral RdRp shows a relation to the reverse transcriptase (RT) enzymes of group II introns that encode RTs and retrotransposons, the latter of which are self-replicating DNA sequences that integrate themselves into other parts of the same DNA molecule. Within the kingdom, +ssRNA viruses are likely to be the oldest lineage, dsRNA viruses appear to have emerged on multiple occasions from +ssRNA viruses, and -ssRNA viruses in turn appear to be related to reoviruses, which are dsRNA viruses.[1][2]
Classification
RNA viruses that encode RdRp are assigned to the kingdom Orthornavirae, which contains five phyla and several taxa that are unassigned to a phylum due to lack of information. The five phyla are separated based on the genome types, host ranges, and genetic similarity of member viruses.[1][20]
- Phylum: Duplornaviricota, which contains dsRNA viruses that infect prokaryotes and eukaryotes, which do not cluster with members of Pisuviricota, and which encode a capsid composed of a 60 homo- or heterodimers of capsid proteins organized on a lattice with pseudo T = 2 symmetry
- Phylum: Kitrinoviricota, which contains +ssRNA viruses that infect eukaryotes and which do not cluster with members of Pisuviricota
- Phylum: Lenarviricota, which contains +ssRNA viruses that infect prokaryotes and eukaryotes and which do not cluster with members of Kitrinoviricota
- Phylum: Negarnaviricota, which contains all -ssRNA viruses[note 2]
- Phylum: Pisuviricota, which contains +ssRNA and dsRNA viruses that infect eukaryotes and which do not cluster with other phyla
The unassigned taxa are listed hereafter (-viridae denotes family and -virus denotes genus).[1][20]
- Birnaviridae
- Permutotetraviridae
- Botybirnavirus
The kingdom contains three groups in the Baltimore classification system, which groups viruses together based on their manner of mRNA synthesis, and which is often used alongside standard virus taxonomy, which is based on evolutionary history. Those three groups are Group III: dsRNA viruses, Group IV: +ssRNA viruses, and Group V: -ssRNA viruses.[1][2]
Disease
RNA viruses are associated with a wide range of disease, including many of the most widely known viral diseases. Notable disease-causing viruses in Orthornavirae include:[20]
- coronaviruses
- Crimean-Congo hemorrhagic fever orthonairovirus
- Dengue virus
- Ebolavirus
- hantaviruses
- Hepatitis A virus
- Hepatitis C virus
- Hepatitis E virus
- Human orthopneumovirus
- influenza viruses
- Japanese encephalitis virus
- Lassa mammarenavirus
- Measles morbillivirus
- Mumps orthorubulavirus
- Norovirus
- Poliovirus
- Rabies lyssavirus
- Rhinoviruses
- Rift Valley fever phlebovirus
- Rotavirus
- Rubella virus
- West Nile virus
- Yellow fever virus
- Zika virus
Animal viruses in Orthornavirae include orbiviruses, which cause various diseases in ruminants and horses, including Bluetongue virus, African horse sickness virus, Equine encephalosis virus, and epizootic hemorrhagic disease virus.[21] The vesicular stomatitis virus causes disease in cattle, horses, and pigs.[22] Bats harbor many viruses including ebolaviruses and henipaviruses, which also can cause disease in humans.[23] Similarly, arthropod viruses in the Flavivirus and Phlebovirus genera are numerous and often transmitted to humans.[24][25] Coronaviruses and influenza viruses cause disease in various vertebrates, including bats, birds, and pigs.[26][27]
Plant viruses in the kingdom are numerous and infect many economically important crops. Tomato spotted wilt virus is estimated to cause more than 1 billion USD in damages annually, affecting more than 800 plant species including chrysanthemum, lettuce, peanut, pepper, and tomato. Cucumber mosaic virus infects more than 1,200 plant species and likewise causes significant crop losses. Potato virus Y causes significant reductions in yield and quality for pepper, potato, tobacco, and tomato, and Plum pox virus is the most important virus among stone fruit crops. Brome mosaic virus, while not causing significant economic losses, is found throughout much of the world and primarily infects grasses, including cereals.[20][28]
History
Diseases caused by RNA viruses in Orthornavirae have been known throughout much of history, but their cause was only discovered in modern times. As a whole, RNA viruses were discovered during a time period of major advancements in molecular biology, including the discovery of mRNA as the immediate carrier of genetic information for protein synthesis.[29] Tobacco mosaic virus was discovered in 1898 and was the first virus to be discovered.[30] Viruses in the kingdom that are transmitted by arthropods have been a key target in the development of vector control, which often aims to prevent viral infections.[31] In modern history, numerous disease outbreaks have been caused by RdRp-encoding RNA viruses, including outbreaks caused by coronaviruses, ebola, and influenza.[32]
Orthornavirae was established in 2019 as a kingdom within the realm Riboviria, intended to accommodate all RdRp-encoding RNA viruses. Prior to 2019, Riboviria was established in 2018 and included only RdRp-encoding RNA viruses. In 2019, Riboviria was expanded to also include reverse transcribing viruses, placed under the kingdom Pararnavirae, so Orthornavirae was established to separate RdRp-encoding RNA viruses from reversing transcribing viruses.[1][33]
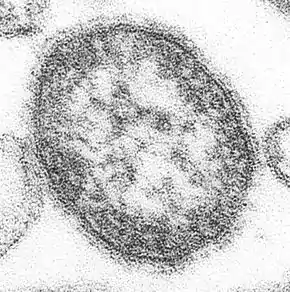
Gallery



_EM_PHIL_2175_lores.jpg.webp)
 Parainfluenza (Paramyxoviridae)
Parainfluenza (Paramyxoviridae)
_EM_18_lores.jpg.webp)
Notes
- The exception is that some members of the order Nidovirales encode proofreading exoribonuclease activity as part of a protein that is distinct from RdRp.
- Excluding deltaviruses, which do not encode RdRp and which therefore are not included in Orthornavirae.
References
- Koonin EV, Dolja VV, Krupovic M, Varsani A, Wolf YI, Yutin N, Zerbini M, Kuhn JH (18 October 2019). "Create a megataxonomic framework, filling all principal taxonomic ranks, for realm Riboviria" (docx). International Committee on Taxonomy of Viruses (ICTV). Retrieved 6 August 2020.
- Wolf YI, Kazlauskas D, Iranzo J, Lucia-Sanz A, Kuhn JH, Krupovic M, Dolja VV, Kooning EV (27 November 2018). "Origins and Evolution of the Global RNA Virome". mBio. 9 (6): e02329-18. doi:10.1128/mBio.02329-18. PMC 6282212. PMID 30482837. Retrieved 6 August 2020.
- "Viral budding". ViralZone. Swiss Institute of Bioinformatics. Retrieved 6 August 2020.
- Fermin, G. (2018). Viruses: Molecular Biology, Host Interactions and Applications to Biotechnology. Elsevier. p. 35-46. doi:10.1016/B978-0-12-811257-1.00002-4. ISBN 9780128112571. Retrieved 6 August 2020.
- "Positive stranded RNA virus replication". ViralZone. Swiss Institute of Bioinformatics. Retrieved 6 August 2020.
- "Subgenomic RNA transcription". ViralZone. Swiss Institute of Bioinformatics. Retrieved 6 August 2020.
- "Negative-stranded RNA virus transcription". ViralZone. Swiss Institute of Bioinformatics. Retrieved 6 August 2020.
- "Negative stranded RNA virus replication". ViralZone. Swiss Institute of Bioinformatics. Retrieved 6 August 2020.
- "Cap snatching". ViralZone. Swiss Institute of Bioinformatics. Retrieved 6 August 2020.
- "Negative-stranded RNA virus polymerase stuttering". ViralZone. Swiss Institute of Bioinformatics. Retrieved 6 August 2020.
- "Ambisense transcription in negative stranded RNA viruses". ViralZone. Swiss Institute of Bioinformatics. Retrieved 6 August 2020.
- "Double-stranded RNA virus replication". ViralZone. Swiss Institute of Bioinformatics. Retrieved 6 August 2020.
- Sanjuán R, Domingo-Calap P (December 2016). "Mechanisms of viral mutation". Cell Mol Life Sci. 73 (23): 4433–4448. doi:10.1007/s00018-016-2299-6. PMC 5075021. PMID 27392606. Retrieved 6 August 2020.
- Smith EC (27 April 2017). "The not-so-infinite malleability of RNA viruses: Viral and cellular determinants of RNA virus mutation rates". PLoS Pathog. 13 (4): e1006254. doi:10.1371/journal.ppat.1006254. PMC 5407569. PMID 28448634. Retrieved 6 August 2020.
- Marsh GA, Rabadan R, Levine AJ, Pelese P (March 2008). "Highly conserved regions of influenza a virus polymerase gene segments are critical for efficient viral RNA packaging". J Virol. 82 (5): 2295–2304. doi:10.1128/JVI.02267-07. PMC 2258914. PMID 18094182. Retrieved 6 August 2020.
- Simon-Loriere E, Holmes EC (4 July 2011). "Why do RNA viruses recombine?". Nat Rev Microbiol. 9 (8): 617–626. doi:10.1038/nrmicro2614. PMC 3324781. PMID 21725337. Retrieved 6 August 2020.
- McDonald SM, Nelson MI, Turner PE, Patton JT (July 2016). "Reassortment in segmented RNA viruses: mechanisms and outcomes". Nat Rev Microbiol. 14 (7): 448–460. doi:10.1038/nrmicro.2016.46. PMC 5119462. PMID 27211789. Retrieved 6 August 2020.
- Vijaykrishna D, Mukerji R, Smith GJ (9 July 2015). "RNA Virus Reassortment: An Evolutionary Mechanism for Host Jumps and Immune Evasion". PLoS Pathog. 11 (7): e1004902. doi:10.1371/journal.ppat.1004902. PMC 4497687. PMID 26158697. Retrieved 6 August 2020.
- Drake JW, Holland JJ (23 November 1999). "Mutation rates among RNA viruses". Proc Natl Acad Sci U S A. 96 (24): 13910–13913. doi:10.1073/pnas.96.24.13910. PMC 24164. PMID 10570172. Retrieved 6 August 2020.
- "Virus Taxonomy: 2019 Release". talk.ictvonline.org. International Committee on Taxonomy of Viruses. Retrieved 6 August 2020.
- Maclachlan NJ, Guthrie AJ (December 2010). "Re-emergence of bluetongue, African horse sickness, and other orbivirus diseases". Vet Res. 41 (6): 35. doi:10.1051/vetres/2010007. PMC 2826768. PMID 20167199. Retrieved 15 August 2020.
- Rozo-Lopez P, Drolet BS, Londono-Renteria B (11 December 2018). "Vesicular Stomatitis Virus Transmission: A Comparison of Incriminated Vectors". Insects. 9 (4): 190. doi:10.3390/insects9040190. PMC 6315612. PMID 30544935. Retrieved 15 August 2020.
- Wang L, Anderson DE (February 2019). "Viruses in bats and potential spillover to animals and humans". Curr Opin Virol. 34: 79–89. doi:10.1016/j.coviro.2018.12.007. PMC 7102861. PMID 30665189.
- Holbrook MR (30 April 2017). "Historical Perspectives on Flavivirus Research". Viruses. 9 (5): 97. doi:10.3390/v9050097. PMC 5454410. PMID 28468299. Retrieved 15 August 2020.
- Hartman A (June 2017). "Rift Valley Fever". Clin Lab Med. 37 (2): 285–301. doi:10.1016/j.cll.2017.01.004. PMC 5458783. PMID 28457351.
- Fehr AR, Perlamn S (2015). "Coronaviruses: an overview of their replication and pathogenesis". Methods Mol Biol. 1282: 1–23. doi:10.1007/978-1-4939-2438-7_1. PMC 4369385. PMID 25720466.
- Webster RG, Govorkova EA (September 2014). "Continuing challenges in influenza". Ann N Y Acad Sci. 1323 (1): 115–139. doi:10.1111/nyas.12462. PMC 4159436. PMID 24891213.
- Scholthof KB, Adkins S, Czosnek H, Palukaitis P, Jacquot E, Hohn T, Hohn B, Saunders K, Candresse T, Ahlquist P, Hemenway C, Foster GD (December 2011). "Top 10 plant viruses in molecular plant pathology". Mol Plant Pathol. 12 (9): 938–954. doi:10.1111/j.1364-3703.2011.00752.x. PMC 6640423. PMID 22017770. Retrieved 6 August 2020.
- Kolakofsky D (April 2015). "A short biased history of RNA viruses". RNA. 21 (4): 667–669. doi:10.1261/rna.049916.115. PMC 4371325. PMID 25780183. Retrieved 6 August 2020.
- Harrison BD, Wilson TM (29 March 1999). "Milestones in the Research on Tobacco Mosaic Virus" (PDF). Philos Trans R Soc Lond B Biol Sci. 354 (1383): 521–529. doi:10.1098/rstb.1999.0403. PMC 1692547. PMID 10212931. Retrieved 6 August 2020.
- Wilson AL, Courtenay O, Kelly-Hope LA, Scott TW, Takken W, Torr SJ, Lindsay SW (16 January 2020). "The Importance of Vector Control for the Control and Elimination of Vector-Borne Diseases". PLoS Negl Trop Dis. 14 (1): e0007831. doi:10.1371/journal.pntd.0007831. PMC 6964823. PMID 31945061. Retrieved 6 August 2020.
- Norris SL, Sawin VI, Ferri M, Sastre LR, Porgo TV (30 May 2018). "An Evaluation of Emergency Guidelines Issued by the World Health Organization in Response to Four Infectious Disease Outbreaks". PLoS One. 13 (5): e0198125. doi:10.1371/journal.pone.0198125. PMC 5976182. PMID 29847593. Retrieved 6 August 2020.
- Gorbalenya, Alexander E.; Krupovic, Mart; Siddell, Stuart; Varsani, Arvind; Kuhn, Jens H. (15 October 2018). "Riboviria: establishing a single taxon that comprises RNA viruses at the basal rank of virus taxonomy" (docx). International Committee on Taxonomy of Viruses (ICTV). Retrieved 6 August 2020.