Histamine trifluoromethyl toluidide
Histamine trifluoromethyl toluidide (HTFMT) is a mixed H1/H2 histamine agonist which is significantly more potent than histamine itself.[1]
 | |
| Names | |
|---|---|
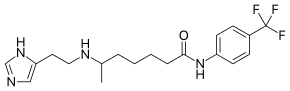
| IUPAC name
6-[2-(1H-imidazol-5-yl)ethylamino]-N-[4-(trifluoromethyl)phenyl]heptanamide | |
| Other names
HTFMT, HTMT | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C19H25F3N4O | |
| Molar mass | 382.42321 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
It also produces additional actions which appear to be independent of histamine receptors.[2][3]
References
- Whyment AD, Blanks AM, Lee K, Renaud LP, Spanswick D. Histamine excites neonatal rat sympathetic preganglionic neurons in vitro via activation of H1 receptors. Journal of Neurophysiology. 2006 Apr;95(4):2492-500. PMID 16354729
- Qiu R, Melmon KL, Khan MM. Effects of histamine-trifluoromethyl-toluidide derivative (HTMT) on intracellular calcium in human lymphocytes. Journal of Pharmacology and Experimental Therapeutics. 1990 Jun;253(3):1245-52. PMID 2359026
- Kim DC, Lee SY, Jun DJ, Kim SH, Lee JH, Hur EM, Baek NI, Kim KT. Inhibition of store-operated calcium entry-mediated superoxide generation by histamine trifluoromethyltoluide independent of histamine receptors. Biochemical Pharmacology. 2005 Nov 25;70(11):1613-22. PMID 16219299
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.