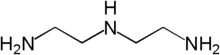
Diethylenetriamine
and also known as 2,2’-Iminodi(ethylamine)[2]) is an organic compound with the formula HN(CH2CH2NH2)2. This colourless hygroscopic liquid is soluble in water and polar organic solvents, but not simple hydrocarbons. Diethylenetriamine is structural analogue of diethylene glycol. Its chemical properties resemble those for ethylene diamine, and it has similar uses. It is a weak base and its aqueous solution is alkaline. DETA is a byproduct of the production of ethylenediamine from ethylene dichloride.[3]
 | |
 | |
 | |
| Names | |
|---|---|
| Other names
N-(2-Aminoethyl)-1,2-ethanediamine; bis(2-Aminoethyl)amine; DETA; 2,2'-Diaminodiethylamine | |
| Identifiers | |
3D model (JSmol) |
|
| 605314 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.003.515 |
| EC Number |
|
| 2392 | |
| MeSH | diethylenetriamine |
PubChem CID |
|
| RTECS number |
|
| UNII | |
| UN number | 2079 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C4H13N3 | |
| Molar mass | 103.169 g·mol−1 |
| Appearance | Colourless liquid |
| Odor | Ammoniacal |
| Density | 955 mg mL−1 |
| Melting point | −39.00 °C; −38.20 °F; 234.15 K |
| Boiling point | 204.1 °C; 399.3 °F; 477.2 K |
| miscible[1] | |
| log P | −1.73 |
| Vapor pressure | 10 Pa (at 20 °C) |
Refractive index (nD) |
1.484 |
| Thermochemistry | |
Heat capacity (C) |
254 J K−1 mol−1 (at 40 °C) |
Std enthalpy of formation (ΔfH⦵298) |
−65.7–−64.7 kJ mol−1 |
Std enthalpy of combustion (ΔcH⦵298) |
−3367.2–−3366.2 kJ mol−1 |
| Hazards | |
| GHS pictograms | 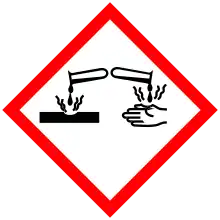  |
| GHS Signal word | Danger |
| H302 | |
| P280, P305+351+338, P310 | |
| Flash point | 102 °C (216 °F; 375 K) |
| 358 °C (676 °F; 631 K) | |
| Explosive limits | 2–6.7% |
| NIOSH (US health exposure limits): | |
PEL (Permissible) |
none[1] |
REL (Recommended) |
TWA 1 ppm (4 mg/m3)[1] |
IDLH (Immediate danger) |
N.D.[1] |
| Related compounds | |
Related amines |
|
Related compounds |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Diethylenetriamine (abbreviated Dien or DETA)
Reactions and uses
Diethylenetriamine is a common curing agent for epoxy resins in epoxy adhesives and other thermosets.[4] It is N-alkylated upon reaction with epoxide groups forming crosslinks.
In coordination chemistry, it serves as a tridentate ligand forming complexes such as Co(dien)(NO2)3.[5]
Like some related amines, it is used in oil industry for the extraction of acid gas.
Like ethylenediamine, DETA can also be used to sensitize nitromethane, making a liquid explosive compound similar to PLX. This compound is cap sensitive with an explosive velocity of around 6200 m/s and is discussed in patent #3,713,915. Mixed with unsymmetrical dimethylhydrazine it was used as Hydyne, a propellent for liquid-fuel rockets.
DETA has been evaluated for use in the Countermine System under development by the U.S. Office of Naval Research, where it would be used to ignite and consume the explosive fill of land mines in beach and surf zones.[6]
See also
References
- NIOSH Pocket Guide to Chemical Hazards. "#0211". National Institute for Occupational Safety and Health (NIOSH).
- "Health Council of the Netherlands: Committee on Updating of Occupational Exposure Limits. 2,2'-Iminodi(ethylamine); Health-based Reassessment of Administrative Occupational Exposure Limits" (PDF). 2005.
- Eller, K.; Henkes, E.; Rossbacher, R.; Höke, H. "Amines, Aliphatic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a02_001.
- Brydson, J. A. (1999). "Epoxide Resins". In J. A. Brydson (ed.). Plastics Materials (Seventh ed.). Oxford: Butterworth-Heinemann. pp. 744–777. doi:10.1016/B978-075064132-6/50067-X. ISBN 9780750641326.
- Crayton, P. H.; Zitomer, F.; Lambert, J. (1963). "Inner Complexes of Cobalt(III) with Diethylenetriamine". In Kleinberg, J. (ed.). Inorganic Syntheses. 7. pp. 207–213. doi:10.1002/9780470132388.ch56. ISBN 9780470132388.
- Hill, Brandon (January 25, 2007). "U.S. Navy Announces "Venom Penetrator" Countermine Projectile". DailyTech. Archived from the original on October 4, 2014. Retrieved July 16, 2013.