ADB-FUBINACA
ADB-FUBINACA is a designer drug identified in synthetic cannabis blends in Japan in 2013.[1] In 2018, it was the third-most common synthetic cannabinoid identified in drugs seized by the Drug Enforcement Administration.[2]
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
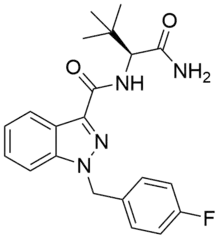
| Formula | C21H23FN4O2 |
| Molar mass | 382.439 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
The (S)-enantiomer of ADB-FUBINACA is described in a 2009 Pfizer patent[3] and has been reported to be a potent agonist of the CB1 receptor and the CB2 receptor with EC50 values of 1.2 nM and 3.5 nM, respectively.[3][4] ADB-FUBINACA features a carboxamide group at the 3-indazole position, like SDB-001 and STS-135. ADB-FUBINACA appears to be the product of rational drug design, since it differs from AB-FUBINACA only by the replacement of the isopropyl group with a tert-butyl group.
An analogue of ADB-FUBINACA, ADSB-FUB-187, containing a more functionalized carboxamide substituent was recently reported.
Side effects
One death through coronary arterial thrombosis has been linked to ADB-FUBINACA intoxication.[5]
At least an additional 8 deaths in Hungary in 2015 are linked to the usage of this material, all deaths were youngsters below 21.
Metabolism
Twenty-three ADB-FUBINACA major metabolites were identified in several incubations with cryopreserved human hepatocytes. Major metabolic pathways were alkyl and indazole hydroxylation, terminal amide hydrolysis, subsequent glucuronide conjugations, and dehydrogenation.[6]
Legality
In the United States, ADB-FUBINACA is a Schedule I controlled substance.[7]
See also
References
- Uchiyama N, Matsuda S, Kawamura M, Kikura-Hanajiri R, Goda Y (July 2013). "Two new-type cannabimimetic quinolinyl carboxylates, QUPIC and QUCHIC, two new cannabimimetic carboxamide derivatives, ADB-FUBINACA and ADBICA, and five synthetic cannabinoids detected with a thiophene derivative α-PVT and an opioid receptor agonist AH-7921 identified in illegal products". Forensic Toxicology. 31 (2): 223–240. doi:10.1007/s11419-013-0182-9. S2CID 1279637.
- "Emerging Threat Report: Annual 2018" (PDF). Special Testing and Research Laboratory, Drug Enforcement Administration.
- WO 2009106982, "Indazole Derivatives"
- Banister SD, Moir M, Stuart J, Kevin RC, Wood KE, Longworth M, et al. (September 2015). "Pharmacology of Indole and Indazole Synthetic Cannabinoid Designer Drugs AB-FUBINACA, ADB-FUBINACA, AB-PINACA, ADB-PINACA, 5F-AB-PINACA, 5F-ADB-PINACA, ADBICA, and 5F-ADBICA". ACS Chemical Neuroscience. 6 (9): 1546–59. doi:10.1021/acschemneuro.5b00112. PMID 26134475.
- Shanks KG, Clark W, Behonick G (April 2016). "Death Associated With the Use of the Synthetic Cannabinoid ADB-FUBINACA". Journal of Analytical Toxicology. 40 (3): 236–9. doi:10.1093/jat/bkv142. PMC 4885918. PMID 26755539.
- Carlier J, Diao X, Wohlfarth A, Scheidweiler K, Huestis MA (July 2017). "In Vitro Metabolite Profiling of ADB-FUBINACA, A New Synthetic Cannabinoid". Current Neuropharmacology. 15 (5): 682–691. doi:10.2174/1570159X15666161108123419. PMC 5771045. PMID 29403341.
- "Schedules of Controlled Substances: Temporary Placement of Six Synthetic Cannabinoids (5F-ADB, 5F-AMB, 5F-APINACA, ADB-FUBINACA, MDMB-CHMICA and MDMB-FUBINACA) Into Schedule I". Drug Enforcement Administration.