Chronic fatigue syndrome
Chronic fatigue syndrome (CFS), also called myalgic encephalomyelitis (ME) and ME/CFS, is a complex, fatiguing, long-term medical condition diagnosed by required primary symptoms and criteria, often involving a broad range of symptoms. Distinguishing core symptoms are lengthy exacerbations or "flares" of the illness after ordinary minor physical or mental activity, known as post-exertional malaise (PEM);[11][12] greatly diminished capacity to accomplish tasks that were routine before the illness; and sleep disturbances.[11][13][1][4][2]:7 Orthostatic intolerance (difficulty sitting and standing upright) and cognitive dysfunction are also diagnostic. Other common symptoms may involve numerous body systems, and chronic pain is common.[13][1]
| Chronic fatigue syndrome | |
|---|---|
| Other names | Myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS),[1] myalgic encephalomyelitis (ME), post-viral fatigue syndrome (PVFS), chronic fatigue immune dysfunction syndrome (CFIDS), systemic exertion intolerance disease (SEID), others[2]:20 |
| Specialty | Primary care, neurology, rheumatology, infectious diseases, physical therapy, occupational therapy, mental health, behavioral health[2]:223 |
| Symptoms | Worsening of symptoms with activity, long-term fatigue, others[1] |
| Usual onset | 40 to 60 years old[3] |
| Duration | Often years[4] |
| Causes | Unknown[1] |
| Risk factors | Female gender, virus and bacterial infections, genetics, major injury, bodily response to severe stress and others[5][6]:1–2 |
| Diagnostic method | Based on symptoms[1] |
| Treatment | Symptomatic[7][8] |
| Frequency | About 0.68 to 1% globally[9][10] |
While the cause is not understood, proposed mechanisms include biological, genetic, infectious, and physical or psychological stress affecting the biochemistry of the body.[5][14] Diagnosis is based on the patient's symptoms because no confirmed diagnostic test is available.[15] The fatigue in CFS is not due to strenuous ongoing exertion, is not much relieved by rest, and is not due to a previous medical condition.[13] Fatigue is a common symptom in many illnesses, but the unexplained fatigue and severity of functional impairment in CFS are relatively rare in these other illnesses.[16]
Persons with CFS may recover or improve over time, but some will become severely affected and disabled for an extended period.[17] No therapies or medications are approved to treat the cause of the illness; treatment is aimed at symptomatology.[7][18] The CDC recommends pacing (personal activity management) to keep mental and physical activity from making symptoms worse.[7] Limited evidence suggests that rintatolimod, counseling, and graded exercise helps some patients.[19]
About 1% of primary-care patients have CFS; estimates of incidence vary widely because epidemiological studies define the illness dissimilarly.[10][15][9] It has been estimated that 836,000 to 2.5 million Americans and 250,000 to 1,250,000 people in the United Kingdom have CFS.[1][20] CFS occurs 1.5 to 2 times as often in women as in men.[10] It most commonly affects adults between ages 40 and 60 years;[3] it can occur at other ages, including childhood.[21] Other studies suggest that about 0.5% of children have CFS, and that it is more common in adolescents than in younger children.[2]:182[21] Chronic fatigue syndrome is a major cause of school absence.[2]:183 CFS reduces health, happiness, and productivity; but there is controversy over many aspects of the disorder. Physicians, researchers, and patient advocates promote different names[22] and diagnostic criteria; and evidence of proposed causes and treatments is often poor or contradictory.[23]
Signs and symptoms
The United States Centers for Disease Control and Prevention (CDC) recommends these criteria for diagnosis:[13]
- Greatly lowered ability to do activities that were usual before the illness. This drop in activity level occurs along with fatigue and must last six months or longer.
- Worsening of symptoms after physical or mental activity that would not have caused a problem before the illness. The amount of activity that might aggravate the illness is difficult for a person to predict, and the decline often presents 12 to 48 hours after the activity.[24] The 'relapse', or 'crash', may last days, weeks or longer. This is known as post-exertional malaise (PEM).
- Sleep problems; people may still feel weary after full nights of sleep, or may struggle to stay awake, fall asleep or stay asleep.
Additionally, one of the following symptoms must be present:[13]
- Problems with thinking and memory (cognitive dysfunction, sometimes described as "brain fog")
- While standing or sitting upright; lightheadedness, dizziness, weakness, fainting or vision changes may occur (orthostatic intolerance)
Other common symptoms
Many, but not all people with ME/CFS report:[13]
- Muscle pain, joint pain without swelling or redness, and headache
- Tender lymph nodes in the neck or armpits
- Sore throat
- Irritable bowel syndrome
- Chills and night sweats
- Allergies and sensitivities to foods, odors, chemicals, lights, or noise
- Shortness of breath
- Irregular heartbeat
The CDC proposes that persons with symptoms resembling those of CFS consult a physician to rule out several treatable illnesses: Lyme disease,[25] "sleep disorders, major depressive disorder, alcohol/substance abuse, diabetes mellitus, hypothyroidism, mononucleosis (mono), lupus, multiple sclerosis (MS), chronic hepatitis and various malignancies."[26] Medications can also cause side effects that mimic symptoms of CFS.[25] Central sensitization, or increased sensitivity to sensory stimuli such as pain have been observed in CFS. Sensitivity to pain increases after exertion, which is opposite to the normal pattern.[27]
Onset
Gradual or sudden onset of the illness may occur, and studies have mixed results as to which occurs more frequently.[2]:158:181
Physical functioning
The functional capacity of individuals with CFS varies greatly.[28] Some persons with CFS lead relatively normal lives; others are totally bed-ridden and unable to care for themselves.[29] For the majority of persons with CFS, work, school, and family activities are significantly reduced for extended periods of time.[30] The severity of symptoms and disability is the same regardless of gender,[31] and many experience strongly disabling chronic pain.[32] Persons report critical reductions in levels of physical activity.[33] Also, a reduction in the complexity of activity has been observed.[34] Reported impairment is comparable to other fatiguing medical conditions[35] including late-stage AIDS,[36] lupus, rheumatoid arthritis, chronic obstructive pulmonary disease (COPD), and end-stage kidney disease.[37] CFS affects a person's functional status and well-being more than major medical conditions such as multiple sclerosis, congestive heart failure, or type II diabetes mellitus.[38][39]
Often, courses of remission and relapse of symptoms occur, which make the illness difficult to manage. Persons who feel better for a period may overextend their activities, and the result can be a worsening of their symptoms with a relapse of the illness.[24]
About 25% of people with CFS are house-bound or bed-ridden for long periods during their illness, often for decades.[2]:32[4] An estimated 75% are unable to work because of their illness.[40] More than half were on disability benefits or temporary sick leave, and less than a fifth worked full-time.[29] Children who become ill with CFS are a major cause of school absence.[2]:183
People with CFS have decreased scores on the SF-36 quality of life questionnaire, especially in the sub scales on vitality, physical functioning, general health, physical role, and social functioning; however, the sub scales for "role emotional" and mental health in CFS patients were consistent with or not substantially lower than healthy controls.[41] Direct healthcare costs are estimated at between $9 and $14 billion annually in the U.S. alone.[40]
Cognitive functioning
Cognitive dysfunction is one of the more disabling aspects of CFS due to its negative impact on occupational and social functioning. 50 to 80 % of persons with CFS are estimated to have serious problems with cognition.[42] Cognitive symptoms are mainly due to deficits in attention, memory, and reaction time. Measured cognitive abilities are found to be below projected normal values and likely to affect day-to-day activities; for example, increases in common mistakes, forgetting scheduled tasks, or having difficulty responding when spoken to are observed.[43]
Simple and complex information-processing speed, and functions entailing working memory over long time periods are moderately to extensively impaired. These deficits are generally consistent with the patient's perceptions. Perceptual abilities, motor speed, language, reasoning, and intelligence do not appear to be significantly altered. When poorer health status was reported, a person's perception of their cognitive problems was frequently greater. Better physical functioning in people with CFS is associated with less visuoperceptual difficulty and fewer language-processing complaints.[43]
Inconsistencies of subjective and observed values of cognitive dysfunction reported across multiple studies are likely caused by a number of factors. Differences of research participants' cognitive abilities pre and post illness onset are naturally variable, and are difficult to measure because of a lack of specialized analytical tools that can consistently quantify the specific cognitive difficulties in CFS.[43]
The frequency of neuropsychiatric and neuropsychological symptoms is increased in the population of persons with CFS; the understanding of why this occurs is unresolved. Various hypotheses have been advanced to try to explain the relationship between the cognitive symptoms and the illness. Some researchers believe psychiatric causes underlie or contribute to the illness, while other researchers believe the illness causes biochemical and sociological changes in people that produce the symptoms.[42]
Cause
The cause of CFS is unknown.[44] Genetic, physiological, and psychological factors are thought to work together to precipitate and perpetuate the condition.[14] A 2016 report by the Institute of Medicine states that CFS is a biologically based illness, but that the biologic abnormalities are not sensitive or specific enough to be useful as a diagnosis.[44]
Because it may begin as an influenza-like illness with a sudden onset, various infectious causes have been proposed, but evidence is insufficient to support such causation.[45][2] Infections proposed include mononucleosis, Chlamydophila pneumoniae, human herpesvirus 6, and Lyme disease. Inflammation may be involved.[46] Often, the illness will follow a viral illness, such as mononucleosis or gastroenteritis.[47]
Risk factors
All ages, ethnic groups, and income levels are susceptible to the illness. The CDC states that Caucasians may be diagnosed more frequently than other races in America,[4] but the illness is at least as prevalent among African Americans and Hispanics.[3] A 2009 meta-analysis showed that compared with Caucasians, African Americans, and Native Americans have a higher risk of CFS, though it specifically excluded other more common ethnicities worldwide, and it acknowledged that studies and data were limited.[48]
More women than men get CFS.[4] A large 2020 meta-analysis estimated that between 1.5 and 2.0 times more cases are women. The review acknowledged that different case definitions and diagnostic methods within datasets yielded a wide range of prevalence rates.[10] The CDC estimates CFS occurs up to four times more often in women than in men.[3] The illness can occur at any age, but most frequently in persons between the ages of 40 and 60.[3] CFS is less prevalent among children and adolescents than among adults.[21]
Blood relatives of those who have CFS appear to be more predisposed, implying that genetic factors may increase the risk of susceptibility to the illness.[12]
Psychological stress, childhood trauma, perfectionist personalities, old age, lower middle education, low physical fitness, preexisting psychological illness, and allergies may be risk factors for developing chronic fatigue syndrome. This has led some to believe that stress-related visceral responses underlie CFS.[49][50] Pre-existing depressive and anxiety disorders, as well as high expectation of parents and family history were predisposing factors identified in another review.[51]
People with CFS and their relatives tend to attribute their illness to physical causes (such as a virus or pollution) rather than to psychological causes,[14][52] and these attributions are associated with increased symptoms and impairment, and worse outcomes over time.[14] According to the CDC, "CFS is a biological illness, not a psychologic disorder", and those affected "are neither malingering nor seeking secondary gain".[53] The World Health Organization (WHO) classifies CFS as a neurological disease in the ICD-11 for Mortality and Morbidity Statistics (ICD-11).[54]
Viral and other infections
The term post-viral fatigue syndrome (PVFS) is used to describe CFS-like symptoms that occur after a viral infection.[6] A recent review found Epstein-Barr Virus (EBV) antibody activity to be higher in patients with CFS, and that a subset of patients with CFS were likely to have increased EBV activity compared to controls.[55] Viral infection is a significant risk factor for CFS, with one study finding 22% of people with Epstein-Barr virus experience fatigue six months later, and 9% having strictly defined CFS.[56] A systematic review found that fatigue severity was the main predictor of prognosis in CFS, and did not identify psychological factors linked to prognosis.[57] One review found risk factors for developing CFS after mononucleosis, dengue fever or the bacterial infection Q-fever include longer bed-rest during the illness, poorer pre-illness physical fitness, attributing symptoms to physical illness, belief that a long recovery time is needed, as well as pre-infection distress and fatigue. The same review found biological factors such as CD4 and CD8 activation and liver inflammation are predictors of sub-acute fatigue, but not CFS,[58] however these findings are not generally accepted due to the use of the Oxford criteria in selecting patients. The CDC does not recognize attribution of symptoms as a risk factor.[5]
A study comparing diagnostic labels found that people labelled with ME had the worst prognosis, while those with PVFS had the best. Whether this is due to those with more severe or longer lasting symptoms results in a label with the description of ME, or if being labelled with ME adversely causes a more severe or prolonged illness is unclear.[59]
Pathophysiology
Neurological
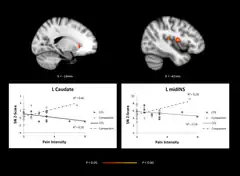
Brain imagining, comparing adolescents with CFS and healthy controls showing abnormal network activity in regions of the brain.A range of neurological structural and functional abnormalities is found in people with CFS, including lowered metabolism at the brain stem, and reduced blood flow to areas of the brain; these differences are consistent with neurological illness, but not depression or psychological illness.[6] The World Health Organization classes chronic fatigue syndrome as a central nervous system disease.[60]
Some neuroimaging studies have observed prefrontal and brainstem hypometabolism; however, sample size was limited.[61] Neuroimaging studies in persons with CFS have identified changes in brain structure, and correlations with various symptoms. Results were not consistent across the neuroimaging brain structure studies, and more research is needed to resolve the discrepancies found between the disparate studies.[62][61]
Tentative evidence suggests a relationship between autonomic nervous system dysfunction and diseases such as CFS, fibromyalgia, irritable bowel syndrome, and interstitial cystitis. However, it is unknown if this relationship is causative.[63] Reviews of CFS literature have found autonomic abnormalities such as decreased sleep efficiency, increased sleep latency, decreased slow wave sleep, and abnormal heart rate response to tilt table tests suggesting a role of the autonomic nervous system in CFS. However, these results were limited by inconsistency.[64][65][66]
Immunological
Immunological abnormalities are frequently observed in those with CFS. Decreased NK cell activity is found more often in people with CFS and this correlates with severity of symptoms.[5][67] People with CFS have an abnormal response to exercise, including increased production of complement products, increased oxidative stress combined with decreased antioxidant response, and increased Interleukin 10, and TLR4, some of which correlates with symptom severity.[68] Increased levels of cytokines have been proposed to account for the decreased ATP production and increased lactate during exercise;[69][70] however, the elevations of cytokine levels are inconsistent in specific cytokine, albeit frequently found.[2][71] Similarities have been drawn between cancer and CFS with regard to abnormal intracellular immunological signaling. Abnormalities observed include hyperactivity of Ribonuclease L, a protein activated by IFN, and hyperactivity of NF-κB.[72]
Endocrine
Evidence points to abnormalities in the hypothalamic-pituitary-adrenal axis (HPA axis) in some, but not all, persons with CFS, which may include slightly low cortisol levels,[73] a decrease in the variation of cortisol levels throughout the day, decreased responsiveness of the HPA axis, and a high serotonergic state, which can be considered to be a "HPA axis phenotype" that is also present in some other conditions, including post-traumatic stress disorder and some autoimmune conditions.[74] It is unclear whether or not decreased cortisol levels of the HPA axis plays a primary role as a cause of CFS,[75][76][77] or has a secondary role in the continuation or worsening of symptoms later in the illness.[78] In most healthy adults, the cortisol awakening response shows an increase in cortisol levels averaging 50% in the first half-hour after waking. In people with CFS, this increase apparently is significantly less, but methods of measuring cortisol levels vary, so this is not certain.[79]
Autoimmunity
Autoimmunity has been proposed to be a factor in CFS, but there are only a few relevant findings so far. A subset of patients with increased B cell activity and autoantibodies, possibly as a result of decreased NK cell regulation or viral mimicry.[80] In 2015, a large German study found 29% of ME/CFS patients had elevated autoantibodies to M3 and M4 muscarinic acetylcholine receptors as well as to ß2 adrenergic receptors.[81][82][83] A 2016 Australian study found that ME/CFS patients had significantly greater numbers of single nucleotide polymorphisms associated with the gene encoding for M3 muscarinic acetylcholine receptors.[84]
Energy metabolism
Studies have observed mitochondrial abnormalities in cellular energy production, but recent focus has concentrated on secondary effects that may result in aberrant mitochondrial function because inherent problems with the mitochondria structure or genetics have not been replicated.[85]
Diagnosis
No characteristic laboratory abnormalities are approved to diagnose CFS; while physical abnormalities can be found, no single finding is considered sufficient for diagnosis.[86][6] Blood, urine, and other tests are used to rule out other conditions that could be responsible for the symptoms.[87][88][2] The CDC states that a medical history should be taken and a mental and physical examination should be done to aid diagnosis.[87]
Diagnostic tools
The CDC recommends considering the questionnaires and tools described in the Institute of Medicine report, which include:
- The Chalder Fatigue Scale
- Multidimensional Fatigue Inventory
- Fisk Fatigue Impact Scale
- The Krupp Fatigue Severity Scale
- DePaul Symptom Questionnaire
- CDC Symptom Inventory for CFS
- Work and Social Adjustment Scale (WSAS)
- SF-36 / RAND-36[2]:270
A two-day cardiopulmonary exercise test (CPET) is not necessary for diagnosis, although lower readings on the second day may be helpful in supporting a claim for social security disability. A two-day CPET cannot be used to rule out chronic fatigue syndrome.[2]:216
Definitions
Notable definitions include:[89]
- Centers for Disease Control and Prevention (CDC) definition (1994),[90] the most widely used clinical and research description of CFS,[14] is also called the Fukuda definition and is a revision of the Holmes or CDC 1988 scoring system.[91] The 1994 criteria require the presence of four or more symptoms beyond fatigue, while the 1988 criteria require six to eight.[92]
- The ME/CFS 2003 Canadian Clinical working definition[93] states: "A patient with ME/CFS will meet the criteria for fatigue, post-exertional malaise and/or fatigue, sleep dysfunction, and pain; have two or more neurological/cognitive manifestations and one or more symptoms from two of the categories of autonomic, neuroendocrine, and immune manifestations; and the illness persists for at least 6 months".
- The Myalgic Encephalomyelitis International Consensus Criteria (ICC) published in 2011 is based on the Canadian working definition and has an accompanying primer for clinicians[94][6] The ICC does not have a six months waiting time for diagnosis. The ICC requires post-exertional neuroimmune exhaustion (PENE) which has similarities with post-exertional malaise, plus at least three neurological symptoms, at least one immune or gastrointestinal or genitourinary symptom, and at least one energy metabolism or ion transportation symptom. Unrefreshing sleep or sleep dysfunction, headaches or other pain, and problems with thinking or memory, and sensory or movement symptoms are all required under the neurological symptoms criterion.[94] According to the ICC, patients with post-exertional neuroimmune exhaustion but only partially meet the criteria should be given the diagnosis of atypical myalgic encephalomyelitis.[6]
- The 2015 definition by the National Academy of Medicine (then referred to as the "Institute of Medicine") is not a definition of exclusion (differential diagnosis is still required).[2] "Diagnosis requires that the patient have the following three symptoms: 1) A substantial reduction or impairment in the ability to engage in pre-illness levels of occupational, educational, social, or personal activities, that persists for more than 6 months and is accompanied by fatigue, which is often profound, is of new or definite onset (not lifelong), is not the result of ongoing excessive exertion, and is not substantially alleviated by rest, and 2) post-exertional malaise* 3) Unrefreshing sleep*; At least one of the two following manifestations is also required: 1) Cognitive impairment* 2) Orthostatic intolerance" and notes that "*Frequency and severity of symptoms should be assessed. The diagnosis of ME/CFS should be questioned if patients do not have these symptoms at least half the time with moderate, substantial, or severe intensity."[2]
Clinical practice guidelines are generally based on case descriptions, with the aim of improving diagnosis, management and treatment. An example is the CFS/ME guideline for the National Health Services in England and Wales, produced in 2007,[92] (presently being updated).[95] Other guidance can be found at the New York Department of Health.[96]
Differential diagnosis
Certain medical conditions can cause chronic fatigue and must be ruled out before a diagnosis of CFS can be given. Hypothyroidism, anemia,[97] coeliac disease (that can occur without gastrointestinal symptoms),[98] diabetes and certain psychiatric disorders are a few of the diseases that must be ruled out if the patient presents with appropriate symptoms.[92][90][97] Other diseases, listed by the Centers for Disease Control and Prevention, include infectious diseases (such as Epstein–Barr virus, influenza, HIV infection, tuberculosis, Lyme disease), neuroendocrine diseases (such as thyroiditis, Addison's disease, adrenal insufficiency, Cushing's disease), hematologic diseases (such as occult malignancy, lymphoma), rheumatologic diseases (such as fibromyalgia, polymyalgia rheumatica, Sjögren's syndrome, giant-cell arteritis, polymyositis, dermatomyositis), psychiatric diseases (such as bipolar disorder, schizophrenia, delusional disorders, dementia, anorexia/bulimia nervosa), neuropsychologic diseases (such as obstructive sleep apnea, parkinsonism, multiple sclerosis), and others (such as nasal obstruction from allergies, sinusitis, anatomic obstruction, autoimmune diseases, some chronic illness, alcohol or substance abuse, pharmacologic side effects, heavy metal exposure and toxicity, marked body weight fluctuation).[97] Ehlers Danlos syndromes (EDS) may also have similar symptoms.[99]
Persons with fibromyalgia (FM, or fibromyalgia syndrome, FMS), like those with CFS, have muscle pain, severe fatigue and sleep disturbances. The presence of allodynia (abnormal pain responses to mild stimulation) and of extensive tender points in specific locations differentiates FM from CFS, although the two diseases often co-occur.[100]
Depressive symptoms, if seen in CFS, may be differentially diagnosed from primary depression by the absence of anhedonia, decreased motivation, and guilt; and the presence of somatic symptoms such as sore throat, swollen lymph nodes, and exercise intolerance with post exertional exacerbation of symptoms.[97]
Management
There is no approved pharmacological treatment, therapy or cure for CFS[7][92] although various drugs have been or are being investigated.[101] A 2014 report prepared by the Agency for Healthcare Research and Quality stated that there are wide variations in patient management, that many receive a multifaceted approach to treatment, and that no medications have been approved by the U.S. Food and Drug Administration (FDA) for the treatment of ME/CFS, although several have been used off label. The report concluded that although counseling and graded exercise therapy (GET) have shown some benefits, these interventions have not been studied fully enough to recommend them for all persons affected. The report expressed concern that GET appears to be associated with worsening symptoms in some.[102] The CDC no longer recommends these interventions, and there is some evidence of patient harm.[103][104]
The CDC guide for the management of CFS states that while there is no cure, a number of methods might improve symptoms.[7] Treatment strategies for sleep problems, pain, (depression, stress, and anxiety) dizziness and lightheadedness (orthostatic Intolerance), and memory and concentration problems are enumerated. Other useful topics mentioned that patients and doctors might discuss include carefully monitoring and managing activity to avoid worsening of symptoms, counseling to cope with the impact the illness may have on quality of life, proper nutrition and nutritional supplements that may support better health, complementary therapies that might help increase energy or decrease pain.[7]
The United Kingdom's National Institute for Health and Clinical Excellence (NICE) 2007 guideline directed toward clinicians, specifies the need for shared decision-making between the patient and healthcare professionals, and acknowledges the reality and impact of the condition and the symptoms. The NICE guideline covers illness management aspects of diet, sleep and sleep disorders, rest, relaxation, and pacing. Referral to specialist care for cognitive behavioural therapy, graded exercise therapy and activity management (pacing) programmes are recommended to be offered as a choice to patients with mild or moderate CFS.[105] In 2017 NICE announced its guidance for CFS/ME needed to be updated,[106] and publication is expected in December 2020.[107]
Comorbid conditions can occur in CFS which may interact with and exacerbate the symptoms of CFS. Appropriate medical intervention for these conditions may be beneficial. The most commonly diagnosed include: fibromyalgia, irritable bowel syndrome, depression, anxiety, as well as allergies and chemical sensitivities.[108]
Pacing
Pacing, or activity management, is an illness management strategy based on the observation that symptoms tend to increase following mental or physical exertion,[7] and was recommended for CFS in the 1980s.[109] It is now commonly used as a management strategy in chronic illnesses and in chronic pain.[110]
Its two forms are: symptom-contingent pacing, where the decision to stop (and rest or change an activity) is determined by a self awareness of an exacerbation of symptoms; and time-contingent pacing, which is determined by a set schedule of activities that a patient estimates he or she is able to complete without triggering postexertional malaise (PEM). Thus, the principle behind pacing for CFS is to avoid overexertion and an exacerbation of symptoms. It is not aimed at treating the illness as a whole. Those whose illness appears stable may gradually increase activity and exercise levels, but according to the principle of pacing, must rest if it becomes clear that they have exceeded their limits.[109] Use of a heart-rate monitor with pacing to monitor and manage activity levels is recommended by a number of patient groups and the UK's 2007 NICE guideline.[111][106]
Energy envelope theory
Energy envelope theory is considered to be consistent with pacing, and is a management strategy suggested in the 2011 international consensus criteria for ME, which referred to using an "energy bank budget". Energy envelope theory was devised by psychologist Leonard Jason, a former sufferer of CFS.[112] Energy envelope theory states that patients should stay within the envelope of energy available to them, and avoid pushing through, which will reduce the postexertional malaise "payback" caused by overexerting and may help them make "modest gains" in physical functioning.[113][114] Several studies have found energy envelope theory to be a helpful management strategy, noting that it reduces symptoms and may increase the level of functioning in CFS.[115][116][114] Energy envelope theory does not recommend unilaterally increasing or decreasing activity and is not intended as a therapy or cure for CFS.[115] It has been promoted by various patient groups.[117][118] Some patient groups recommend using a heart rate monitor to increase awareness of exertion, and allow patients to stay within their aerobic threshold envelope.[119][120] Despite a number of studies showing positive results for energy envelope theory, randomized controlled trials are lacking.
Exercise
Stretching, movement therapies, and toning exercises are recommended for pain in patients with CFS, and pain medication is also suggested. In many chronic illnesses, aerobic exercise is beneficial, but in chronic fatigue syndrome, the CDC does not recommend it. The CDC states:[7]
"Any activity or exercise plan for people with ME/CFS needs to be carefully designed with input from each patient. While vigorous aerobic exercise can be beneficial for many chronic illnesses, patients with ME/CFS do not tolerate such exercise routines. Standard exercise recommendations for healthy people can be harmful for patients with ME/CFS. However, it is important that patients with ME/CFS undertake activities that they can tolerate..."
Counseling
The CDC states that counseling may help patients cope with pain caused by CFS, and that talking with a professional counselor or therapist may help people to more effectively manage the symptoms that affect their quality of daily life.[7]
Nutrition
A proper diet is a significant contributor to the health of any individual. Medical consultation about diet and supplements are recommended for persons with CFS.[7] Persons with CFS may benefit from a balanced diet and properly supervised administration of nutritional support if deficiencies are detected by medical testing. Risks of nutritional supplements include interactions with prescribed medications.[121][7]
Therapies
Cognitive behavioral therapy
The CDC states that speaking with a therapist may help people cope with the illness.[7] A 2015 National Institutes of Health report concluded that while counseling and behavior therapies could produce benefits for some people, they may not yield improvement in quality of life, and because of this limitation such therapies should not be considered as a primary treatment, but rather should be used only as one component of a broader approach.[122] This same report stated that although counseling approaches have shown benefit in some measures of fatigue, function and overall improvement, these approaches have been inadequately studied in subgroups of the wider CFS patient population. Further concern was expressed that reporting of negative effects experienced by patients receiving counseling and behavior therapies had been poor.[102] A report by the Institute of Medicine published in 2015 states that it is unclear whether CBT helps to improve cognitive impairments experienced by patients.[2]:265 The rationale behind the use of CBT to change beliefs about the illness is disputed.[103]
A 2008 Cochrane Review concluded that CBT did reduce the symptom of fatigue, but noted that the benefits of CBT may diminish after the therapy is completed, and that due to study limitations "the significance of these findings should be interpreted with caution".[23] A 2014 systematic review reported that there was only limited evidence that patients increased levels of physical activity after receiving CBT. The authors concluded that, as this finding is contrary to the cognitive behavioural model of CFS, patients receiving CBT were adapting to the illness rather than recovering from it.[123]
Patient organisations have long criticised the use of CBT as a treatment for CFS, and the rationale behind the model is disputed.[104][124] In 2012 the ME Association (MEA) commenced an opinion survey of 493 patients who had received a CBT treatment in the UK. Based on the finding of this survey, in 2015 the MEA concluded that CBT in its current form should not be recommended as a primary intervention for people with CFS[125] In a letter published online in the Lancet in 2016, Dr Charles Shepherd, medical advisor to the MEA, expressed the view that the contention between patients and researchers lay in "a flawed model of causation that takes no account of the heterogeneity of both clinical presentations and disease pathways that come under the umbrella diagnosis of ME/CFS".[126] In 2019, a large UK survey of people with ME/CFS reported that CBT was ineffective for more than half of people, and Graded Exercise Therapy caused deterioration in most people.[127]
Graded exercise therapy
Previously, a 2014 National Institutes of Health report concluded that while graded exercise therapy (GET) could produce benefits, it may not yield improvement in quality of life and because of this limitation, GET should not be considered as a primary treatment, but instead be used only as one component of a broader approach. The report also noted that a focus on exercise programs had discouraged patient participation in other types of physical activity, due to concerns of precipitating increased symptoms.[122] A July 2016 addendum to this report recommended that the Oxford criteria not be used when studying ME/CFS. If studies based on the Oxford criteria were excluded, there would be insufficient evidence of the effectiveness of GET on any outcome.[104]
A 2002 Cochrane review updated in 2019 stated that exercise therapy probably has a positive effect on fatigue in adults, and slightly improves sleep, but the long-term effects are unknown, and this has limited relevance to current definitions of ME/CFS.[128][8] Cochrane have announced that a new review to look at exercise therapies in chronic fatigue syndrome is to start in 2020.[8][129] As with CBT, patient organisations have long criticised the use of exercise therapy, most notably GET, as a treatment for CFS.[124] In 2012 the MEA commenced an opinion survey of patients who had received GET. Based on the findings of this survey, in 2015 the MEA concluded that GET in its current delivered form should not be recommended as a primary intervention for persons with CFS.[125]
Adaptive pacing therapy
Adaptive pacing therapy (APT) was popularised by the PACE trial, a study that has caused much controversy among both patients and practitioners.[19] APT, not to be confused with pacing,[130] is a therapy rather than a management strategy.[131] APT is based on the idea that CFS involves a person only having a limited amount of available energy, and using this energy wisely will mean the "limited energy will increase gradually".[131]:5 A large clinical trial known as the PACE trial found APT was no more effective than usual care or specialized medical care.[132] Unlike pacing, APT is based on the cognitive behavioral model of chronic fatigue syndrome and involves increasing activity levels, which it states may temporarily increase symptoms.[133] In APT, the patient first establishes a baseline level of activity, which can be carried out consistently without any postexertional malaise ("crashes"). APT states that persons should plan to increase their activity, as able. However, APT also requires patients to restrict their activity level to only 70% of what they feel able to do, while also warning against too much rest.[131] This has been described as contradictory, and Jason states that in comparison with pacing, this 70% limit restricts the activities that patients are capable of and results in a lower level of functioning.[130] Jason and Goudsmit, who first described pacing and the energy envelope theory for CFS, have both criticized APT for being inconsistent with the principles of pacing and highlighted significant differences.[130] APT was promoted by Action for ME, the patient charity involved in the PACE trial, until 2019.[133]
Rintatolimod
Rintatolimod is a double-stranded RNA drug developed to modulate an antiviral immune reaction through activation of toll-like receptor 3. In several clinical trials of CFS, the treatment has shown a reduction in symptoms, but improvements were not sustained after discontinuation.[134] Evidence supporting the use of rintatolimod is deemed low to moderate.[19] The US FDA has denied commercial approval, called a new drug application, citing several deficiencies and gaps in safety data in the trials, and concluded that the available evidence is insufficient to demonstrate its safety or efficacy in CFS.[135][136] Rintatolimod has been approved for marketing and treatment for persons with CFS in Argentina,[137] and in 2019, FDA regulatory requirements were met for exportation of rintatolimod to the country.[138]
Prognosis
A systematic review which looked at the course of CFS without systematic biological or psychological interventions found that "the median full recovery rate was 5% (range 0–31%) and the median proportion of patients who improved during follow-up was 39.5% (range 8–63%). Return to work at follow-up ranged from 8 to 30% in the three studies that considered this outcome." ... "In five studies, a worsening of symptoms during the period of follow-up was reported in between 5 and 20% of patients." A good outcome was associated with not attributing illness to a physical cause, and having a sense of control over symptoms. Other factors were occasionally, but not consistently, related to outcome, including age at onset, a longer duration of follow-up, and less fatigue severity at baseline. The review concludes that "irrespective of the biology of CFS, patients’ beliefs and attributions about the illness are intricately linked with the clinical presentation, the type of help sought and prognosis"[139] Another review found that children have a better prognosis than adults, with 54–94% having recovered by follow-up compared to less than 10% of adults returning to pre-illness levels of functioning.[140]
Epidemiology
The prevalence rates for CFS/ME vary widely depending on "case definitions and diagnostic methods".[10] Based on the 1994 CDC diagnostic criteria, the global prevalence rate for CFS is 0.89%.[10] In comparison, the prevalence rate for the stricter criteria, such as the 1988 CDC "Holmes" criteria for CFS and the 2003 Canadian criteria for ME (both of which, for example, exclude patients with psychiatric diagnoses), produce an incidence rate of only 0.17%.[10] For an example of how these rates impact a nation: the CDC website states that "836,000 to 2.5 million Americans suffer from ME/CFS", but most remain undiagnosed.[1]
Females are diagnosed about 1.5 to 2.0 times more often with CFS than males.[10] An estimated 0.5% of children have CFS, and more adolescents are affected with the illness than younger children.[2]:182[21]
History
Myalgic encephalomyelitis
- From 1934 onwards, outbreaks of a previously unknown illness began to be recorded by doctors.[141][142] Initially considered to be occurrences of poliomyelitis, the illness was subsequently referred to as "epidemic neuromyasthenia".[142]
- In the 1950s, the term "benign myalgic encephalomyelitis" was used in relation to a comparable outbreak at the Royal Free Hospital in London.[143] The descriptions of each outbreak were varied, but included symptoms of malaise, tender lymph nodes, sore throat, pain, and signs of encephalomyelitis.[144] The cause of the condition was not identified, although it appeared to be infectious, and the term "benign myalgic encephalomyelitis" was chosen to reflect the lack of mortality, the severe muscular pains, symptoms suggesting damage to the nervous system, and to the presumed inflammatory nature of the disorder. Björn Sigurðsson disapproved of the name, stating that the illness is rarely benign, doesn't always cause muscle pain, and is possibly never encephalomyelitic.[141] The syndrome appeared in sporadic as well as epidemic cases.[145]
- In 1969, benign myalgic encephalomyelitis appeared as an entry to the International Classification of Diseases under Diseases of the nervous system.[146]
- In 1986, Ramsay published the first diagnostic criteria for ME, in which the condition was characterized by: 1) muscle fatiguability in which, even after minimal physical effort, 3 or more days elapse before full muscle power is restored; 2) extraordinary variability or fluctuation of symptoms, even in the course of one day; and 3) chronicity.[147]
- By 1988, the continued work of Ramsay had demonstrated that, although the disease rarely resulted in mortality, it was often severely disabling.[2]:28–29 Because of this, Ramsay proposed that the prefix "benign" be dropped.[143][148][149]
Chronic fatigue syndrome
- In the mid-1980s, two large outbreaks of an illness that resembled mononucleosis drew national attention in the United States. Located in Nevada and New York, the outbreaks involved an illness characterized by "chronic or recurrent debilitating fatigue, and various combinations of other symptoms, including a sore throat, lymph node pain and tenderness, headache, myalgia, and arthralgias". An initial link to the Epstein-Barr virus had the illness acquire the name "chronic Epstein-Barr virus syndrome".[2]:29[91]
- In 1987, the CDC convened a working group to reach a consensus on the clinical features of the illness. The working group concluded that CFS was not new, and that the many different names given to it previously reflected widely differing concepts of the illness's cause and epidemiology.[150] The CDC working group chose "chronic fatigue syndrome" as a more neutral and inclusive name for the illness, but noted that "myalgic encephalomyelitis" was widely accepted in other parts of the world.[91]
- In 1988, the first definition of CFS was published. Although the cause of the illness remained unknown, several attempts were made to update this definition, most notably in 1994.[90]
- The most widely referenced diagnostic criteria and definition of CFS for research and clinical purposes were published in 1994 by the CDC.[59]
- In 2006, the CDC commenced a national program to educate the American public and health-care professionals about CFS.[151]
Other medical terms
A range of both theorised and confirmed medical entities and naming conventions have appeared historically in the medical literature dealing with ME and CFS. These include:
- Epidemic neuromyasthenia was a term used for outbreaks with symptoms resembling poliomyelitis.[141][152]
- Iceland disease and Akureyri disease were synonymous terms used for an outbreak of fatigue symptoms in Iceland.[153]
- Low natural killer syndrome, a term used mainly in Japan, reflected research showing diminished in vitro activity of natural killer cells isolated from patients.[154][155]
- Neurasthenia has been proposed as an historical diagnosis that occupied a similar medical and cultural space to CFS.[156]
- Royal Free disease was named after the historically significant outbreak in 1955 at the Royal Free Hospital used as an informal synonym for "benign myalgic encephalomyelitis".[157]
- Tapanui flu was a term commonly used in New Zealand, deriving from the name of a town, Tapanui, where numerous people had the syndrome.[158]
Society and culture

Naming
Many names have been proposed for the illness. Currently, the most commonly used are "chronic fatigue syndrome", "myalgic encephalomyelitis", and the umbrella term "ME/CFS". Reaching consensus on a name is challenging because the cause and pathology remain unknown.[2]:29–30
The term "chronic fatigue syndrome" has been criticized by some patients as being both stigmatizing and trivializing, and which in turn prevents the illness from being seen as a serious health problem that deserves appropriate research.[159] While many patients prefer "myalgic encephalomyelitis", which they believe better reflects the medical nature of the illness,[147][160] there is resistance amongst some clinicians toward the use of myalgic encephalomyelitis on the grounds that the inflammation of the central nervous system (myelitis) implied by the term has not been demonstrated.[161][162]
A 2015 report from the Institute of Medicine recommended the illness be renamed "systemic exertion intolerance disease", (SEID), and suggested new diagnostic criteria, proposing post-exertional malaise, (PEM), impaired function, and sleep problems are core symptoms of ME/CFS. Additionally, they described cognitive impairment and orthostatic intolerance as distinguishing symptoms from other fatiguing illnesses.[2][163][164]
Economic impact
Reynolds et al. (2004)[165] estimated that the illness caused about $20,000 per person with CFS in lost productivity, which totals to $9.1 billion per year in the United States.[166] This is comparable to other chronic illnesses that extract some of the biggest medical and socioeconomic costs.[167] A 2008 study[168] calculated that the total annual cost burden of ME/CFS to society in the US was extensive, and could approach $24.0 billion.[169] A 2017 estimate for the annual economic burden in the United Kingdom from ME/CFS was 3.3 billion Pounds Sterling.[12]
Awareness day
May 12 is designated as ME/CFS International Awareness Day.[170] The day is observed so that stakeholders have an occasion to improve the knowledge of "the public, policymakers, and health-care professionals about the symptoms, diagnosis, and treatment of ME/CFS, as well as the need for a better understanding of this complex illness."[171] It was chosen because it is the birthday of Florence Nightingale, who had an illness appearing similar to ME/CFS or fibromyalgia.[170][172]
Doctor–patient relations
Some in the medical community do not recognize CFS as a real condition, nor does agreement exist on its prevalence.[173][174][175] There has been much disagreement over proposed causes, diagnosis, and treatment of the illness.[176][177][178][179][180] This uncertainty can significantly affect doctor-patient relations. A 2006 survey of GPs in southwest England found that despite more than two-thirds of them accepting CFS/ME as a recognizable clinical entity, nearly half did not feel confident with making the diagnosis and/or treating the disease. Three other key factors that were significantly, positively associated with GPs' attitudes were knowing someone socially with CFS/ME, being male, and seeing more patients with the condition in the last year.[181]
From the patient perspective, one 1997 study found that 77% of individuals with CFS reported negative experiences with health-care providers.[38] In a more recent metaanalysis of qualitative studies, a major theme identified in patient discourses was that they felt severely ill, yet were blamed and dismissed.[182] A study of themes in patient newsgroup postings noted key themes relating to denial of social recognition of suffering and feelings of being accused of "simply faking it". Another theme that emerged strongly was that achieving diagnosis and acknowledgement requires tremendous amounts of "hard work" by patients.[175][183]
Blood donation
In 2010, several national blood banks adopted measures to discourage or prohibit individuals diagnosed with CFS from donating blood, based on concern following the 2009 claim of a link,[184] between CFS and a retrovirus which was subsequently shown to be unfounded. Organizations adopting these or similar measures included the Canadian Blood Services,[185] the New Zealand Blood Service,[186] the Australian Red Cross Blood Service[187] and the American Association of Blood Banks,[188] In November 2010, the UK National Blood Service introduced a permanent deferral of donation from ME/CFS patients based on the potential harm to those patients that may result from their giving blood.[189] Donation policy in the UK now states, "The condition is relapsing by nature and donation may make symptoms worse, or provoke a relapse in an affected individual."[190]
Controversy
Much contention has arisen over the cause, pathophysiology,[50] nomenclature,[191] and diagnostic criteria of CFS.[176][177] Historically, many professionals within the medical community were unfamiliar with CFS, or did not recognize it as a real condition; nor did agreement exist on its prevalence or seriousness.[174][175][192] Some people with CFS reject any psychological component.[193]
Two British psychiatrists, in 1970, reviewed the case notes of 15 outbreaks of benign myalgic encephalomyelitis and concluded that it was caused by mass hysteria on the part of patients, or altered medical perception of the attending physicians.[194][195] Their conclusions were based on previous studies that found many normal physical test results, a lack of a discernible cause, and a higher prevalence of the illness in females. Consequently, the authors recommended that the disease should be renamed "myalgia nervosa". Despite strong refutation by Dr. Melvin Ramsay and other medical professionals, the proposed psychological hypothesis created great controversy, and convinced a generation of health professionals in the UK that this could be a plausible explanation for the condition, resulting in neglect by many medical specialties. The specialty that did take a major interest in the illness was psychiatry.[195]
Because of the controversy, sociologists hypothesized that stresses of modern living might be a cause of the illness, while some in the media used the term "Yuppie flu" and called it a disease of the middle class. People with disabilities from CFS were often not believed and were accused of being malingerers.[195] The November 1990 issue of Newsweek ran a cover story on CFS, which although supportive of an organic cause of the illness, also featured the term 'yuppie flu', reflecting the stereotype that CFS mainly affected yuppies. The implication was that CFS is a form of burnout. The term 'yuppie flu' is considered offensive by both patients and clinicians.[196][197]
In 2009, the journal Science[184] published a study that identified the XMRV retrovirus in a population of people with CFS. Other studies failed to reproduce this finding,[198][199][200] and in 2011, the editor of Science formally retracted its XMRV paper[201] while the Proceedings of the National Academy of Sciences similarly retracted a 2010 paper which had appeared to support the finding of a connection between XMRV and CFS.[202]
United Kingdom
The lack of research funding and the funding bias towards biopsychosocial studies and against biomedical studies has been highlighted a number of times by patient groups and a number of UK politicians.[203] A parliamentary inquiry by an ad hoc group of parliamentarians in the United Kingdom, set up and chaired by former MP, Dr Ian Gibson, called the Group on Scientific Research into CFS/ME,[106]:169–186[204] was addressed by a government minister claiming that few good biomedical research proposals have been submitted to the Medical Research Council (MRC) in contrast to those for psychosocial research. They were also told by other scientists of proposals that have been rejected, with claims of bias against biomedical research. The MRC confirmed to the group that from April 2003 to November 2006, it has turned down 10 biomedical applications relating to CFS/ME and funded five applications relating to CFS/ME, mostly in the psychiatric/psychosocial domain.
In 2008, the MRC set up an expert group to consider how the MRC might encourage new high-quality research into CFS/ME and partnerships between researchers already working on CFS/ME and those in associated areas. It currently lists CFS/ME with a highlight notice, inviting researchers to develop high-quality research proposals for funding.[205] In February 2010, the All-Party Parliamentary Group on ME (APPG on ME) produced a legacy paper, which welcomed the recent MRC initiative, but felt that far too much emphasis in the past had been on psychological research, with insufficient attention to biomedical research, and that further biomedical research must be undertaken to help discover a cause and more effective forms of management for this disease.[206]
Controversy surrounds psychologically oriented models of the disease and behavioral treatments conducted in the UK.[207]
United States
In 1998, $13 million for CFS research was found to have been redirected or improperly accounted for by the United States CDC, and officials at the agency misled Congress about the irregularities. The agency stated that they needed the funds to respond to other public-health emergencies. The director of a U.S. national patient advocacy group charged the CDC had a bias against studying the disease. The CDC pledged to improve their practices and to restore the $13 million to CFS research over three years.[208]
On 29 October 2015, the National Institutes of Health declared its intent to increase research on ME/CFS. The NIH Clinical Center was to study individuals with ME/CFS, and the National Institute of Neurological Disorders and Stroke would lead the Trans-NIH ME/CFS Research Working Group as part of a multi-institute research effort.[209]
Notable cases
In 1989, The Golden Girls (1985–1992) featured chronic fatigue syndrome in a two-episode arc, "Sick and Tired: Part 1" and "Part 2," in which protagonist Dorothy Zbornak, portrayed by Bea Arthur, after a lengthy battle with her doctors in an effort to find a diagnosis for her symptoms, is finally diagnosed with CFS.[210] American author Ann Bannon had CFS.[211] Laura Hillenbrand, author of the popular book Seabiscuit, has struggled with CFS since age 19.[212][213]
Research
The different case definitions used to research the illness influence the types of patients selected for studies,[86] and research also suggests subtypes of patients may exist within a heterogeneous population.[166][214][215][216] In one of the definitions, symptoms are accepted that may suggest a psychiatric disorder, while others specifically exclude primary psychiatric disorders.[89] The lack of a single, unifying case definition was criticized in the Institute of Medicine's 2015 report for "creating an unclear picture of the symptoms and signs of the disorder" and "complicating comparisons of the results" (study results).[2]:72
References
- "Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) | CDC". www.cdc.gov. 2020-04-13. Retrieved 2020-05-20.
- Committee on the Diagnostic Criteria for Myalgic Encephalomyelitis/Chronic Fatigue Syndrome; Board on the Health of Select Populations; Institute of, Medicine (10 February 2015). Beyond Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Redefining an Illness (PDF). PMID 25695122.
- "Epidemiology | Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) | CDC". www.cdc.gov. 2018-07-12. Retrieved 2020-05-24.
- "What is ME/CFS? | Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) | CDC". www.cdc.gov. 2018-07-12. Retrieved 2020-05-21.
- "Possible Causes | Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) | CDC". www.cdc.gov. 15 May 2019. Retrieved 20 May 2020.
- Carruthers BM, van de Sande MI, De Meirleir KL, Klimas NG, Broderick G, Mitchell T, Staines D, Powles ACP, Speight N, Vallings R, Bateman L, Bell DS, Carlo-Stella N, Chia J, Darragh A, Gerken A, Jo D, Lewis D, Light AR, Light K, Marshall-Gradisnik S, McLaren-Howard J, Mena I, Miwa K, Murovska M, Steven S (2012). MYALGIC ENCEPHALOMYELITIS – Adult & Paediatric: International Consensus Primer for Medical Practitioners Authors - International Consensus Panel.
- "Treatment of ME/CFS | Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) | CDC". www.cdc.gov. 2019-11-19. Retrieved 2020-05-22.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - Cochrane (21 May 2020). "Publication of Cochrane Review: 'Exercise therapy for chronic fatigue syndrome'". www.cochrane.org. Retrieved 2020-05-24.
It now places more emphasis on the limited applicability of the evidence to definitions of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) used in the included studies, the long-term effects of exercise on symptoms of fatigue, and acknowledges the limitations of the evidence about harms that may occur.
- Sandler, Carolina X; Lloyd, Andrew R (2020). "Chronic fatigue syndrome: progress and possibilities". Medical Journal of Australia. 212 (9): 428–433. doi:10.5694/mja2.50553. ISSN 0025-729X. PMID 32248536. S2CID 214810583.
- Lim EJ, Ahn YC, Jang ES, Lee SW, Lee SH, Son CG (February 2020). "Systematic review and meta-analysis of the prevalence of chronic fatigue syndrome/myalgic encephalomyelitis (CFS/ME)". J Transl Med. 18 (1): 100. doi:10.1186/s12967-020-02269-0. PMC 7038594. PMID 32093722.
- "Information for Healthcare Providers | CDC". www.cdc.gov. 2020-04-13. Retrieved 2020-06-17.
- Dibble, Joshua J; McGrath, Simon J; Ponting, Chris P (2020-09-30). "Genetic risk factors of ME/CFS: a critical review". Human Molecular Genetics. 29 (R1): R117–R124. doi:10.1093/hmg/ddaa169. PMC 7530519. PMID 32744306.
- "Symptoms of ME/CFS | Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) | CDC". www.cdc.gov. 2019-11-19. Retrieved 2020-05-20.
- Afari N, Buchwald D (February 2003). "Chronic fatigue syndrome: a review". The American Journal of Psychiatry. 160 (2): 221–36. doi:10.1176/appi.ajp.160.2.221. PMID 12562565.
- Estévez-López, Fernando; Mudie, Kathleen; Wang-Steverding, Xia; Bakken, Inger Johanne; Ivanovs, Andrejs; Castro-Marrero, Jesús; Nacul, Luis; Alegre, Jose; Zalewski, Paweł; Słomko, Joanna; Strand, Elin Bolle; Pheby, Derek; Shikova, Evelina; Lorusso, Lorenzo; Capelli, Enrica; Sekulic, Slobodan; Scheibenbogen, Carmen; Sepúlveda, Nuno; Murovska, Modra; Lacerda, Eliana (2020-05-21). "Systematic Review of the Epidemiological Burden of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Across Europe: Current Evidence and EUROMENE Research Recommendations for Epidemiology". Journal of Clinical Medicine. MDPI AG. 9 (5): 1557. doi:10.3390/jcm9051557. ISSN 2077-0383. PMC 7290765. PMID 32455633.
- Ranjith G (January 2005). "Epidemiology of chronic fatigue syndrome". Occupational Medicine. 55 (1): 13–9. doi:10.1093/occmed/kqi012. PMID 15699086.
- "Severely Affected Patients - Clinical Care of Patients - Healthcare Providers - Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS)". CDC. 2019-11-19. Retrieved 2020-11-30.
- "1 Guidance - Chronic fatigue syndrome/myalgic encephalomyelitis (or encephalopathy): diagnosis and management - Guidance". NICE. 2007-08-22. Retrieved 2020-07-11.
- Smith ME, Haney E, McDonagh M, Pappas M, Daeges M, Wasson N, Fu R, Nelson HD (June 2015). "Treatment of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: A Systematic Review for a National Institutes of Health Pathways to Prevention Workshop". Annals of Internal Medicine (Systematic review). 162 (12): 841–50. doi:10.7326/M15-0114. PMID 26075755.
- "Annex 1: Epidemiology of CFS/ME". UK Department of Health. 2012-01-06. Archived from the original on 2012-01-06. Retrieved July 28, 2017.
- "ME/CFS in Children | Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) | CDC". www.cdc.gov. 2019-05-15. Retrieved 2020-05-24.
ME/CFS is often thought of as a problem in adults, but children (both adolescents and younger children) can also get ME/CFS.
- Ottati VC (2002). The social psychology of politics. New York: Kluwer Academic/Plenum. pp. 159–160. ISBN 978-0-306-46723-3. Retrieved 2009-08-11.
- Price JR, Mitchell E, Tidy E, Hunot V (July 2008). Price JR (ed.). "Cognitive behaviour therapy for chronic fatigue syndrome in adults". The Cochrane Database of Systematic Reviews (3): CD001027. doi:10.1002/14651858.CD001027.pub2. PMC 7028002. PMID 18646067.
- "Treating the Most Disruptive Symptoms First and Preventing Worsening of Symptoms | CDC". www.cdc.gov. 2019-11-19. Retrieved 2020-08-19.
- "CDC — Chronic Fatigue Syndrome (CFS) — Diagnosis". Cdc.gov. Retrieved 2012-07-22.
- "CDC, Chronic Fatigue Syndrome (CFS), Making a Diagnosis" (PDF). Cdc.gov. Retrieved 2011-01-28.
- Nijs J, Meeus M, Van Oosterwijck J, Ickmans K, Moorkens G, Hans G, De Clerck LS (February 2012). "In the mind or in the brain? Scientific evidence for central sensitisation in chronic fatigue syndrome". European Journal of Clinical Investigation. 42 (2): 203–12. doi:10.1111/j.1365-2362.2011.02575.x. PMID 21793823. S2CID 13926525.
- Vanness JM, Snell CR, Strayer DR, Dempsey L, Stevens SR (June 2003). "Subclassifying chronic fatigue syndrome through exercise testing". Medicine and Science in Sports and Exercise. 35 (6): 908–13. doi:10.1249/01.MSS.0000069510.58763.E8. PMID 12783037.
- Ross SD, Estok RP, Frame D, Stone LR, Ludensky V, Levine CB (May 2004). "Disability and chronic fatigue syndrome: a focus on function". Archives of Internal Medicine. 164 (10): 1098–107. doi:10.1001/archinte.164.10.1098. PMID 15159267.
- "Presentation and Clinical Course of ME/CFS | Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) | CDC". www.cdc.gov. 2019-11-19. Retrieved 2020-07-11.
- Ho-Yen DO, McNamara I (August 1991). "General practitioners' experience of the chronic fatigue syndrome". The British Journal of General Practice. 41 (349): 324–6. PMC 1371754. PMID 1777276.
- Meeus M, Nijs J, Meirleir KD (May 2007). "Chronic musculoskeletal pain in patients with the chronic fatigue syndrome: a systematic review". European Journal of Pain. 11 (4): 377–86. doi:10.1016/j.ejpain.2006.06.005. PMID 16843021. S2CID 21414690.
- McCully KK, Sisto SA, Natelson BH (January 1996). "Use of exercise for treatment of chronic fatigue syndrome". Sports Medicine. 21 (1): 35–48. doi:10.2165/00007256-199621010-00004. PMID 8771284. S2CID 239650.
- Burton C, Knoop H, Popovic N, Sharpe M, Bleijenberg G (June 2009). "Reduced complexity of activity patterns in patients with chronic fatigue syndrome: a case control study". BioPsychoSocial Medicine. 3 (1): 7. doi:10.1186/1751-0759-3-7. PMC 2697171. PMID 19490619.
- Solomon L, Nisenbaum R, Reyes M, Papanicolaou DA, Reeves WC (October 2003). "Functional status of persons with chronic fatigue syndrome in the Wichita, Kansas, population". Health and Quality of Life Outcomes. 1 (1): 48. doi:10.1186/1477-7525-1-48. PMC 239865. PMID 14577835.
- Mark, Loveless, MD, congressional testimony of, May 12, 1995, as reported in Hillary Johnson. (1996). Osler's Web: Inside the Labyrinth of the Chronic Fatigue Syndrome Epidemic. Crown Publishers, New York. ISBN 0-517-70353-X. pp.364-365
- "Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS)". Centers for Disease Control and Prevention. 13 April 2020.
- Anderson JS, Ferrans CE (June 1997). "The quality of life of persons with chronic fatigue syndrome". The Journal of Nervous and Mental Disease. 185 (6): 359–67. doi:10.1097/00005053-199706000-00001. PMID 9205421.
- Komaroff AL, Fagioli LR, Doolittle TH, Gandek B, Gleit MA, Guerriero RT, Kornish RJ, Ware NC, Ware JE, Bates DW (September 1996). "Health status in patients with chronic fatigue syndrome and in general population and disease comparison groups". The American Journal of Medicine. 101 (3): 281–90. doi:10.1016/S0002-9343(96)00174-X. PMID 8873490.
- "Chronic Fatigue Syndrome: Advancing Research and Clinical Education". Centers for Disease Control and Prevention. 28 February 2018.
- Unger ER, Lin JS, Brimmer DJ, Lapp CW, Komaroff AL, Nath A, Laird S, Iskander J (December 2016). "CDC Grand Rounds: Chronic Fatigue Syndrome - Advancing Research and Clinical Education" (PDF). MMWR. Morbidity and Mortality Weekly Report. 65 (50–51): 1434–1438. doi:10.15585/mmwr.mm655051a4. PMID 28033311.
- Christley, Y; Duffy, T; Everall, IP; Martin, CR (2013). "The neuropsychiatric and neuropsychological features of chronic fatigue syndrome: revisiting the enigma". Current Psychiatry Reports. 15 (4): 353. doi:10.1007/s11920-013-0353-8. ISSN 1523-3812. PMID 23440559. S2CID 25790262.
- Cvejic, Erin; Birch, Rachael C.; Vollmer-Conna, Uté (2016-03-31). "Cognitive Dysfunction in Chronic Fatigue Syndrome: a Review of Recent Evidence". Current Rheumatology Reports. Springer Science and Business Media LLC. 18 (5): 24. doi:10.1007/s11926-016-0577-9. ISSN 1523-3774. PMID 27032787. S2CID 38748839.
- Unger ER, Lin JS, Brimmer DJ, Lapp CW, Komaroff AL, Nath A, Laird S, Iskander J (December 2016). "CDC Grand Rounds: Chronic Fatigue Syndrome - Advancing Research and Clinical Education". MMWR. Morbidity and Mortality Weekly Report. 65 (50–51): 1434–1438. doi:10.15585/mmwr.mm655051a4. PMID 28033311.
- Rasa S, Nora-Krukle Z, Henning N, Eliassen E, Shikova E, Harrer T, Scheibenbogen C, Murovska M, Prusty BK (October 2018). "Chronic viral infections in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS)". J Transl Med. 16 (1): 268. doi:10.1186/s12967-018-1644-y. PMC 6167797. PMID 30285773.
- Gerwyn M, Maes M (January 2017). "Mechanisms Explaining Muscle Fatigue and Muscle Pain in Patients with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS): a Review of Recent Findings". Current Rheumatology Reports. 19 (1): 1. doi:10.1007/s11926-017-0628-x. PMID 28116577. S2CID 11823204.
- "Chronic fatigue syndrome (Tapanui flu)". www.southerncross.co.nz Southern Cross NZ. March 2020. Retrieved 3 February 2021.
- Dinos S, Khoshaba B, Ashby D, White PD, Nazroo J, Wessely S, Bhui KS (December 2009). "A systematic review of chronic fatigue, its syndromes and ethnicity: prevalence, severity, co-morbidity and coping". International Journal of Epidemiology. 38 (6): 1554–70. doi:10.1093/ije/dyp147. PMID 19349479.
- Van Houdenhove B, Kempke S, Luyten P (June 2010). "Psychiatric aspects of chronic fatigue syndrome and fibromyalgia". Current Psychiatry Reports. 12 (3): 208–14. doi:10.1007/s11920-010-0105-y. PMID 20425282. S2CID 19669971.
- Hempel S, Chambers D, Bagnall AM, Forbes C (July 2008). "Risk factors for chronic fatigue syndrome/myalgic encephalomyelitis: a systematic scoping review of multiple predictor studies". Psychological Medicine. 38 (7): 915–26. doi:10.1017/S0033291707001602. PMID 17892624.
- Lievesley K, Rimes KA, Chalder T (April 2014). "A review of the predisposing, precipitating and perpetuating factors in Chronic Fatigue Syndrome in children and adolescents". Clinical Psychology Review (Submitted manuscript). 34 (3): 233–48. doi:10.1016/j.cpr.2014.02.002. PMID 24632047.
- Cho HJ, Hotopf M, Wessely S (2005). "The placebo response in the treatment of chronic fatigue syndrome: a systematic review and meta-analysis". Psychosomatic Medicine. 67 (2): 301–13. doi:10.1097/01.psy.0000156969.76986.e0. PMID 15784798. S2CID 33633322.
- "Myalgic Encephalomyelitis/Chronic Fatigue Syndrome — Etiology and Pathophysiology". 2018-07-10.
- "Mortality and Morbidity Statistics". ICD-11. Retrieved 2021-01-09.
- Eriksen, Willy (16 August 2018). "ME/CFS, case definition, and serological response to Epstein-Barr virus. A systematic literature review". Fatigue: Biomedicine, Health & Behavior. 6 (4): 220–234. doi:10.1080/21641846.2018.1503125. S2CID 80898744.
- Cleare AJ (March 2004). "The HPA axis and the genesis of chronic fatigue syndrome". Trends in Endocrinology and Metabolism. 15 (2): 55–9. doi:10.1016/j.tem.2003.12.002. PMID 15036250. S2CID 1353041.
- Jason, Leonard A.; Porter, Nicole; Brown, Molly; Anderson, Valerie; Brown, Abigail; Hunnell, Jessica; Lerch, Athena (2009). "CFS: A Review of Epidemiology and Natural History Studies". Bulletin of the IACFS/ME. 17 (3): 88–106. PMC 3021257. PMID 21243091.
- Hulme, Katrin; Hudson, Joanna L.; Rojczyk, Philine; Little, Paul; Moss-Morris, Rona (August 2017). "Biopsychosocial risk factors of persistent fatigue after acute infection: A systematic review to inform interventions" (PDF). Journal of Psychosomatic Research. 99: 120–129. doi:10.1016/j.jpsychores.2017.06.013. PMID 28712416.
- Brurberg KG, Fønhus MS, Larun L, Flottorp S, Malterud K (February 2014). "Case definitions for chronic fatigue syndrome/myalgic encephalomyelitis (CFS/ME): a systematic review". BMJ Open. 4 (2): e003973. doi:10.1136/bmjopen-2013-003973. PMC 3918975. PMID 24508851.
- "ICD-11 - Mortality and Morbidity Statistics". icd.who.int. Retrieved 20 May 2020.
Diseases of the nervous system
- Maksoud R, du Preez S, Eaton-Fitch N, Thapaliya K, Barnden L, Cabanas H, Staines D, Marshall-Gradisnik S (2020). "A systematic review of neurological impairments in myalgic encephalomyelitis/ chronic fatigue syndrome using neuroimaging techniques". PLOS ONE. 15 (4): e0232475. Bibcode:2020PLoSO..1532475M. doi:10.1371/journal.pone.0232475. PMC 7192498. PMID 32353033.
- Jason LA, Zinn ML, Zinn MA (2 February 2017). "Myalgic Encephalomyelitis: Symptoms and Biomarkers". Current Neuropharmacology. 13 (5): 701–34. doi:10.2174/1570159X13666150928105725. PMC 4761639. PMID 26411464.
Decreased frontal grey matter
- Martínez-Martínez LA, Mora T, Vargas A, Fuentes-Iniestra M, Martínez-Lavín M (April 2014). "Sympathetic nervous system dysfunction in fibromyalgia, chronic fatigue syndrome, irritable bowel syndrome, and interstitial cystitis: a review of case-control studies". Journal of Clinical Rheumatology. 20 (3): 146–50. doi:10.1097/RHU.0000000000000089. PMID 24662556. S2CID 23799955.
- Jackson ML, Bruck D (December 2012). "Sleep abnormalities in chronic fatigue syndrome/myalgic encephalomyelitis: a review". Journal of Clinical Sleep Medicine. 8 (6): 719–28. doi:10.5664/jcsm.2276. PMC 3501671. PMID 23243408.
- Tanaka M, Tajima S, Mizuno K, Ishii A, Konishi Y, Miike T, Watanabe Y (November 2015). "Frontier studies on fatigue, autonomic nerve dysfunction, and sleep-rhythm disorder". The Journal of Physiological Sciences. 65 (6): 483–98. doi:10.1007/s12576-015-0399-y. PMC 4621713. PMID 26420687.
- Van Cauwenbergh D, Nijs J, Kos D, Van Weijnen L, Struyf F, Meeus M (May 2014). "Malfunctioning of the autonomic nervous system in patients with chronic fatigue syndrome: a systematic literature review". European Journal of Clinical Investigation. 44 (5): 516–26. doi:10.1111/eci.12256. PMID 24601948. S2CID 9722415.
- Lapp, Charles W. (16 February 2016). "Chronic Fatigue Syndrome: Advancing Research and Clinical Education" (PDF). CDC Public Health Grand Rounds. Centers for Disease Control and Prevention.
- Nijs J, Nees A, Paul L, De Kooning M, Ickmans K, Meeus M, Van Oosterwijck J (2014). "Altered immune response to exercise in patients with chronic fatigue syndrome/myalgic encephalomyelitis: a systematic literature review". Exercise Immunology Review. 20: 94–116. PMID 24974723.
- Armstrong, Christopher W.; McGregor, Neil R.; Butt, Henry L.; Gooley, Paul R. (2014). Metabolism in Chronic Fatigue Syndrome. Advances in Clinical Chemistry. 66. pp. 121–172. doi:10.1016/B978-0-12-801401-1.00005-0. ISBN 978-0-12-801401-1. PMID 25344988.
- Morris G, Anderson G, Galecki P, Berk M, Maes M (March 2013). "A narrative review on the similarities and dissimilarities between myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and sickness behavior". BMC Medicine. 11: 64. doi:10.1186/1741-7015-11-64. PMC 3751187. PMID 23497361.
- Griffith JP, Zarrouf FA (2008). "A systematic review of chronic fatigue syndrome: don't assume it's depression". Primary Care Companion to the Journal of Clinical Psychiatry. 10 (2): 120–8. doi:10.4088/pcc.v10n0206. PMC 2292451. PMID 18458765.
- Meeus M, Mistiaen W, Lambrecht L, Nijs J (November 2009). "Immunological similarities between cancer and chronic fatigue syndrome: the common link to fatigue?". Anticancer Research. 29 (11): 4717–26. PMID 20032425.
- Silverman MN, Heim CM, Nater UM, Marques AH, Sternberg EM (May 2010). "Neuroendocrine and immune contributors to fatigue". PM & R. 2 (5): 338–46. doi:10.1016/j.pmrj.2010.04.008. PMC 2933136. PMID 20656615.
- Morris G, Anderson G, Maes M (November 2017). "Hypothalamic-Pituitary-Adrenal Hypofunction in Myalgic Encephalomyelitis (ME)/Chronic Fatigue Syndrome (CFS) as a Consequence of Activated Immune-Inflammatory and Oxidative and Nitrosative Pathways". Molecular Neurobiology. 54 (9): 6806–6819. doi:10.1007/s12035-016-0170-2. PMID 27766535. S2CID 3524276.
- Cho HJ, Skowera A, Cleare A, Wessely S (January 2006). "Chronic fatigue syndrome: an update focusing on phenomenology and pathophysiology". Current Opinion in Psychiatry. 19 (1): 67–73. doi:10.1097/01.yco.0000194370.40062.b0. PMID 16612182. S2CID 12815707.
- Papadopoulos AS, Cleare AJ (September 2011). "Hypothalamic-pituitary-adrenal axis dysfunction in chronic fatigue syndrome". Nature Reviews. Endocrinology. 8 (1): 22–32. doi:10.1038/nrendo.2011.153. PMID 21946893. S2CID 22176725.
- Tak LM, Cleare AJ, Ormel J, Manoharan A, Kok IC, Wessely S, Rosmalen JG (May 2011). "Meta-analysis and meta-regression of hypothalamic-pituitary-adrenal axis activity in functional somatic disorders". Biological Psychology. 87 (2): 183–94. doi:10.1016/j.biopsycho.2011.02.002. PMID 21315796. S2CID 206108463.
- Van Den Eede F, Moorkens G, Van Houdenhove B, Cosyns P, Claes SJ (2007). "Hypothalamic-pituitary-adrenal axis function in chronic fatigue syndrome". Neuropsychobiology. 55 (2): 112–20. CiteSeerX 10.1.1.626.9632. doi:10.1159/000104468. PMID 17596739. S2CID 14956850.
- Powell DJ, Liossi C, Moss-Morris R, Schlotz W (November 2013). "Unstimulated cortisol secretory activity in everyday life and its relationship with fatigue and chronic fatigue syndrome: a systematic review and subset meta-analysis". Psychoneuroendocrinology. 38 (11): 2405–22. doi:10.1016/j.psyneuen.2013.07.004. PMID 23916911.
- Morris G, Berk M, Galecki P, Maes M (April 2014). "The emerging role of autoimmunity in myalgic encephalomyelitis/chronic fatigue syndrome (ME/cfs)". Molecular Neurobiology. 49 (2): 741–56. doi:10.1007/s12035-013-8553-0. hdl:11343/219795. PMID 24068616. S2CID 13185036.
- Loebel, Madlen; Grabowski, Patricia; Heidecke, Harald; Bauer, Sandra; Hanitsch, Leif G.; Wittke, Kirsten; Meisel, Christian; Reinke, Petra; Volk, Hans-Dieter (Feb 2016). "Antibodies to β adrenergic and muscarinic cholinergic receptors in patients with Chronic Fatigue Syndrome". Brain, Behavior, and Immunity. 52: 32–39. doi:10.1016/j.bbi.2015.09.013. ISSN 1090-2139. PMID 26399744.
- "Myalgic Encephalomyelitis/Chronic Fatigue Syndrome – Evidence for an autoimmune disease". Autoimmunity Reviews. 17 (6): 601–609. doi:10.1016/j.autrev.2018.01.009. ISSN 1568-9972. Retrieved 2021-01-29.
- "A Unifying Hypothesis of the Pathophysiology of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS): Recognitions from the finding of autoantibodies against ß2-adrenergic receptors". Autoimmunity Reviews. 19 (6). 2020-06-01. doi:10.1016/j.autrev.2020.102527. ISSN 1568-9972. Retrieved 2021-01-29.
- Marshall-Gradisnik, Sonya; Smith, Peter; Nilius, Bernd; Staines, Donald R. (2015-01-01). "Examination of Single Nucleotide Polymorphisms in Acetylcholine Receptors in Chronic Fatigue Syndrome Patients". Immunology and Immunogenetics Insights. 7: III.S25105. doi:10.4137/III.S25105. ISSN 1178-6345.
- Holden, Sean; Maksoud, Rebekah; Eaton-Fitch, Natalie; Cabanas, Hélène; Staines, Donald; Marshall-Gradisnik, Sonya (2020-02-18). "A systematic review of mitochondrial abnormalities in myalgic encephalomyelitis/chronic fatigue syndrome/systemic exertion intolerance disease". Journal of Translational Medicine. 18 (1): 290. doi:10.1186/s12967-020-02452-3. PMC 7392668. PMID 32727475.
- Reeves WC, Lloyd A, Vernon SD, Klimas N, Jason LA, Bleijenberg G, Evengard B, White PD, Nisenbaum R, Unger ER (December 2003). "Identification of ambiguities in the 1994 chronic fatigue syndrome research case definition and recommendations for resolution". BMC Health Services Research. 3 (1): 25. doi:10.1186/1472-6963-3-25. PMC 317472. PMID 14702202.
- "Diagnosis of ME/CFS | Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) | CDC". 15 May 2019.
- Bansal AS (July 2016). "Investigating unexplained fatigue in general practice with a particular focus on CFS/ME". BMC Family Practice. 17 (81): 81. doi:10.1186/s12875-016-0493-0. PMC 4950776. PMID 27436349.
- Wyller VB (2007). "The chronic fatigue syndrome--an update". Acta Neurologica Scandinavica. Supplementum. 187: 7–14. doi:10.1111/j.1600-0404.2007.00840.x. PMID 17419822. S2CID 11247547.
- Fukuda K, Straus SE, Hickie I, Sharpe MC, Dobbins JG, Komaroff A (December 1994). "The chronic fatigue syndrome: a comprehensive approach to its definition and study. International Chronic Fatigue Syndrome Study Group". Annals of Internal Medicine. 121 (12): 953–9. doi:10.7326/0003-4819-121-12-199412150-00009. PMID 7978722. S2CID 510735.
- Holmes GP, Kaplan JE, Gantz NM, Komaroff AL, Schonberger LB, Straus SE, Jones JF, Dubois RE, Cunningham-Rundles C, Pahwa S (March 1988). "Chronic fatigue syndrome: a working case definition". Annals of Internal Medicine. 108 (3): 387–9. doi:10.7326/0003-4819-108-3-387. PMID 2829679.
- Guideline 53: Chronic fatigue syndrome/myalgic encephalomyelitis (or encephalopathy). London: National Institute for Health and Clinical Excellence. 2007. ISBN 978-1-84629-453-2.
- Carruthers, Bruce M.; Jain, Anil Kumar; De Meirleir, Kenny L.; Peterson, Daniel L.; Klimas, Nancy G.; Lerner, A. Martin; Bested, Alison C.; Flor-Henry, Pierre; Joshi, Pradip; Powles, A. C. Peter; Sherkey, Jeffrey A.; van de Sande, Marjorie I. (4 December 2011). "Myalgic Encephalomyelitis/Chronic Fatigue Syndrome". Journal of Chronic Fatigue Syndrome. 11 (1): 7–115. doi:10.1300/J092v11n01_02.
- Carruthers BM, van de Sande MI, De Meirleir KL, Klimas NG, Broderick G, Mitchell T, et al. (October 2011). "Myalgic encephalomyelitis: International Consensus Criteria". Journal of Internal Medicine. 270 (4): 327–38. doi:10.1111/j.1365-2796.2011.02428.x. PMC 3427890. PMID 21777306.
- "NICE to begin review of its guidance on the diagnosis and treatment of CFS/ME". NICE. 20 September 2017. Archived from the original on 21 April 2018.
- "Myalgic Encephalomyelitis ("Chronic Fatigue Syndrome")". NY DOH diseases and conditions. April 2018.
- Craig T, Kakumanu S (March 2002). "Chronic fatigue syndrome: evaluation and treatment". American Family Physician. 65 (6): 1083–90. PMID 11925084.
- Logan AC, Wong C (October 2001). "Chronic fatigue syndrome: oxidative stress and dietary modifications" (PDF). Alternative Medicine Review. 6 (5): 450–9. PMID 11703165.
Finally, recent evidence suggests celiac disease can present with neurological symptoms in the absence of gastrointestinal symptoms; therefore, celiac disease should be included in the differential diagnosis of CFS.
- Hakim, Alan; De Wandele, Inge; O'Callaghan, Chris; Pocinki, Alan; Rowe, Peter (March 2017). "Chronic fatigue in Ehlers-Danlos syndrome-Hypermobile type". American Journal of Medical Genetics Part C: Seminars in Medical Genetics. 175 (1): 175–180. doi:10.1002/ajmg.c.31542. PMID 28186393. Lay summary.
- Bradley LA, McKendree-Smith NL, Alarcón GS (2000). "Pain complaints in patients with fibromyalgia versus chronic fatigue syndrome". Current Review of Pain. 4 (2): 148–57. doi:10.1007/s11916-000-0050-2. PMID 10998728. S2CID 2555977.
- Smith ME, Nelson HD, Haney E, Pappas M, Daeges M, Wasson N, McDonagh M (December 2014). "Diagnosis and Treatment of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome". Evidence Report/Technology Assessment (Evidence Reports/Technology Assessments, No. 219 ed.). Agency for Healthcare Research and Quality (US) (219): 1–433. doi:10.23970/AHRQEPCERTA219. PMID 30313001. Retrieved 22 January 2016.
- Smith ME, Nelson HD, Haney E, Pappas M, Daeges M, Wasson N, McDonagh M (December 2014). "Diagnosis and Treatment of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome". Evidence Report/Technology Assessment (Evidence Reports/Technology Assessments, No. 219 ed.). Agency for Healthcare Research and Quality (US) (219): 1–433. doi:10.23970/AHRQEPCERTA219. PMID 30313001. Retrieved 22 January 2016.
- Vink M, Vink-Niese A (2018-07-01). "Graded exercise therapy for myalgic encephalomyelitis/chronic fatigue syndrome is not effective and unsafe. Re-analysis of a Cochrane review". Health Psychology Open. 5 (2): 2055102918805187. doi:10.1177/2055102918805187. PMC 6176540. PMID 30305916.
The analysis of the 2017 Cochrane review reveals flaws, which means that contrary to its findings, there is no evidence that graded exercise therapy is effective. Because of the failure to report harms adequately in the trials covered by the review, it cannot be said that graded exercise therapy is safe. The analysis of the objective outcomes in the trials provides sufficient evidence to conclude that graded exercise therapy is an ineffective treatment for myalgic encephalomyelitis/chronic fatigue syndrome... The analysis of the 2017 Cochrane review reveals flaws, which means that contrary to its findings, there is no evidence that graded exercise therapy is effective. Because of the failure to report harms adequately in the trials covered by the review, it cannot be said that graded exercise therapy is safe. The analysis of the objective outcomes in the trials provides sufficient evidence to conclude that graded exercise therapy is an ineffective treatment for myalgic encephalomyelitis/chronic fatigue syndrome.
- Smith ME, Nelson HD, Haney E, Pappas M, Daeges M, Wasson N, McDonagh M (July 2016). "July 2016 Addendum. In Diagnosis and Treatment of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome 2014 Dec". Evidence Report/Technology Assessment. Agency for Healthcare Research and Quality (US): 1–433.
The results are consistent across trials with improvement in function, fatigue, and global improvement and provided moderate strength of evidence for improved function (4 trials, n=607) and global improvement (3 trials, n=539), low strength of evidence for reduced fatigue (4 trials, n=607) and decreased work impairment (1 trial, n=480), and insufficient evidence for improved quality of life (no trials)
- London: National Institute for Health and Clinical Excellence (2007). Guideline 53: Chronic fatigue syndrome/myalgic encephalomyelitis (or encephalopathy). ISBN 978-1846294532. Retrieved 22 January 2016.
- Chronic fatigue syndrome/myalgic encephalomyelitis (or encephalopathy): diagnosis and management. London: National Institute for Health and Clinical Excellence. 22 August 2007. Retrieved 3 February 2021.
- Myalgic encephalomyelitis (or encephalopathy)/chronic fatigue syndrome: diagnosis and management. London: National Institute for Health and Clinical Excellence. 2018. Retrieved 14 December 2019.
- "Comorbid Conditions | Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) | CDC". www.cdc.gov. 2018-07-12. Retrieved 2020-05-29.
- Goudsmit EM, Nijs J, Jason LA, Wallman KE (19 December 2011). "Pacing as a strategy to improve energy management in myalgic encephalomyelitis/chronic fatigue syndrome: a consensus document". Disability and Rehabilitation. 34 (13): 1140–7. doi:10.3109/09638288.2011.635746. PMID 22181560. S2CID 22457926. Retrieved 2020-05-23.
- Nielson WR, Jensen MP, Karsdorp PA, Vlaeyen JW (May 2013). "Activity pacing in chronic pain: concepts, evidence, and future directions". The Clinical Journal of Pain. 29 (5): 461–8. doi:10.1097/AJP.0b013e3182608561. PMID 23247005. S2CID 28709499.
- "ME Association Summary Review: Assessing PEM (Post-exertional Malaise)" (PDF). ME Association. 2019.
- Jason LA, Melrose H, Lerman A, Burroughs V, Lewis K, King CP, Frankenberry EL (January 1999). "Managing chronic fatigue syndrome: overview and case study". AAOHN Journal. 47 (1): 17–21. doi:10.1177/216507999904700104. PMID 10205371.
- Jason LA, Brown M, Brown A, Evans M, Flores S, Grant-Holler E, Sunnquist M (January 2013). "Energy Conservation/Envelope Theory Interventions to Help Patients with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome". Fatigue. 1 (1–2): 27–42. doi:10.1080/21641846.2012.733602. PMC 3596172. PMID 23504301.
- O'connor K, Sunnquist M, Nicholson L, Jason LA, Newton JL, Strand EB (March 2019). "Energy envelope maintenance among patients with myalgic encephalomyelitis and chronic fatigue syndrome: Implications of limited energy reserves". Chronic Illness. 15 (1): 51–60. doi:10.1177/1742395317746470. PMC 5750135. PMID 29231037.
- Jason L, Muldowney K, Torres-Harding S (May 2008). "The Energy Envelope Theory and myalgic encephalomyelitis/chronic fatigue syndrome". AAOHN Journal. 56 (5): 189–95. doi:10.3928/08910162-20080501-06. PMID 18578185. S2CID 25558691.
- Brown M, Khorana N, Jason LA (March 2011). "The role of changes in activity as a function of perceived available and expended energy in nonpharmacological treatment outcomes for ME/CFS". Journal of Clinical Psychology. 67 (3): 253–60. doi:10.1002/jclp.20744. PMC 3164291. PMID 21254053.
- Campbell, B (Winter 2009). "Managing your energy envelope" (PDF). The CFIDS Chronicle: 28–31.
- "Pacing - Emerge Australia". Emerge Australia. Retrieved 2020-05-23.
- Steefel, Lorraine (2011-09-15). What Nurses Know...Chronic Fatigue Syndrome. Demos Medical Publishing. pp. 54–55. ISBN 978-1-61705-028-2.
- Campbell, Bruce (14 November 2009). "Pacing by Numbers: using your heart rate to stay inside the energy envelope". ME/CFS South Australia Inc. Retrieved 2020-05-23.
- Castro-Marrero J, Sáez-Francàs N, Santillo D, Alegre J (March 2017). "Treatment and management of chronic fatigue syndrome/myalgic encephalomyelitis: all roads lead to Rome". Br. J. Pharmacol. 174 (5): 345–369. doi:10.1111/bph.13702. PMC 5301046. PMID 28052319.
- Green CR, Cowan P, Elk R, O'Neil KM, Rasmussen AL (June 2015). "National Institutes of Health Pathways to Prevention Workshop: Advancing the Research on Myalgic Encephalomyelitis/Chronic Fatigue Syndrome". Annals of Internal Medicine. 162 (12): 860–5. doi:10.7326/M15-0338. PMID 26075757.
- Adamowicz JL, Caikauskaite I, Friedberg F (November 2014). "Defining recovery in chronic fatigue syndrome: a critical review". Quality of Life Research. 23 (9): 2407–16. doi:10.1007/s11136-014-0705-9. PMID 24791749. S2CID 13609292.
- Clark C, Buchwald D, MacIntyre A, Sharpe M, Wessely S (January 2002). "Chronic fatigue syndrome: a step towards agreement". Lancet. 359 (9301): 97–8. doi:10.1016/S0140-6736(02)07336-1. PMID 11809249. S2CID 38526912.
- The ME Association. "No decisions about me without me" (PDF). ME Association. The ME Association. Retrieved 20 January 2016.
- Shepherd C (February 2016). "Patient reaction to the PACE trial". The Lancet. Psychiatry. 3 (2): e7-8. doi:10.1016/S2215-0366(15)00546-5. PMID 26795759. Retrieved 20 January 2016.
- "Forward-ME and Oxford Brookes University announces results of patient survey on CBT and GET in ME/CFS". ME Association. Apr 2019.
- Larun L, Brurberg KG, Odgaard-Jensen J, Price JR (October 2019). "Exercise therapy for chronic fatigue syndrome". The Cochrane Database of Systematic Reviews. 10: CD003200. doi:10.1002/14651858.CD003200.pub8. PMC 6953363. PMID 31577366.
- Elgot J (18 October 2015), "Chronic fatigue patients criticise study that says exercise can help", The Guardian, retrieved 20 June 2018
- Jason LA (August 2017). "The PACE trial missteps on pacing and patient selection". Journal of Health Psychology. 22 (9): 1141–1145. doi:10.1177/1359105317695801. PMID 28805518.
- Cox D, Ludlam S, Mason L, Wagner S, Sharpe M, et al. (PACE Trial Management Group) (November 2004). "Manual for Participants Adaptive Pacing Therapy (APT) for CFS/ME" (PDF). Wolfson Institute | Queen Mary University of London. Retrieved 23 May 2020.
- White PD, Goldsmith KA, Johnson AL, Potts L, Walwyn R, DeCesare JC, et al. (March 2011). "Comparison of adaptive pacing therapy, cognitive behaviour therapy, graded exercise therapy, and specialist medical care for chronic fatigue syndrome (PACE): a randomised trial". Lancet. 377 (9768): 823–36. doi:10.1016/S0140-6736(11)60096-2. PMC 3065633. PMID 21334061.
- "Pacing for people with ME" (PDF). Action for ME. 2013. Archived from the original (PDF) on 5 November 2018.
- Richman S, Morris MC, Broderick G, Craddock TJ, Klimas NG, Fletcher MA (May 2019). "Pharmaceutical Interventions in Chronic Fatigue Syndrome: A Literature-based Commentary". Clin Ther. 41 (5): 798–805. doi:10.1016/j.clinthera.2019.02.011. PMC 6543846. PMID 30871727.
- Research, Center for Drug Evaluation and. "FDA Response Letter Regarding Approval of Ampligen for ME/CFS". www.fda.gov. Archived from the original on 2016-10-23. Retrieved 2018-06-12.
- Barclay, Laura (5 February 2013). "FDA Nixes Rintatolimod for Chronic Fatigue Syndrome". Medscape. Retrieved 18 January 2017.
- Advances in Nucleic Acid Therapeutics. Drug Discovery. RSC Publishing. 2019-02-11. p. 310. doi:10.1039/9781788015714. ISBN 978-1-78801-209-6. Retrieved 2020-05-26.
- "Rintatolimod for severe Chronic Fatigue Syndrome". fda.gov. 19 September 2019. Retrieved 2020-05-26.
- Cairns R, Hotopf M (January 2005). "A systematic review describing the prognosis of chronic fatigue syndrome". Occupational Medicine. 55 (1): 20–31. doi:10.1093/occmed/kqi013. PMID 15699087.
- Joyce J, Hotopf M, Wessely S (March 1997). "The prognosis of chronic fatigue and chronic fatigue syndrome: a systematic review". QJM. 90 (3): 223–33. doi:10.1093/qjmed/90.3.223. PMID 9093600.
- Acheson ED (April 1959). "The clinical syndrome variously called benign myalgic encephalomyelitis, Iceland disease and epidemic neuromyasthenia". The American Journal of Medicine. 26 (4): 569–95. CiteSeerX 10.1.1.534.4761. doi:10.1016/0002-9343(59)90280-3. PMID 13637100.
- Parish JG (November 1978). "Early outbreaks of 'epidemic neuromyasthenia'". Postgraduate Medical Journal. 54 (637): 711–7. doi:10.1136/pgmj.54.637.711. PMC 2425322. PMID 370810.
- Wojcik W, Armstrong D, Kanaan R (June 2011). "Chronic fatigue syndrome: labels, meanings and consequences". Journal of Psychosomatic Research. 70 (6): 500–4. doi:10.1016/j.jpsychores.2011.02.002. PMID 21624573.
- Lancet. Public health (1955). "Outbreak at the royal free". The Lancet. 266 (6885): 351–352. doi:10.1016/s0140-6736(55)92344-8.
- Price JL (April 1961). "Myalgic encephalomyelitis". Lancet. 1 (7180): 737–8. doi:10.1016/s0140-6736(61)92893-8. PMC 1836797. PMID 13737972.
- International Classification of Diseases. I. World Health Organization. 1969. pp. 158, (vol 2, pp. 173).
- Ramsay AM. Myalgic encephalomyelitis and postviral fatigue states. Second Ed. 1988
- Ramsey, A.Melvin; David, Anthonys.; Wessely, Simon; Pelosi, Anthonyj.; Dowsett, E.G. (July 1988). "Myalgic encephalomyelitis, or what?". Lancet. 2 (8602): 100–1. doi:10.1016/s0140-6736(88)90028-1. PMID 2898668. S2CID 24860444.
- Ramsay AM, Dowsett EG, Dadswell JV, Lyle WH, Parish JG (May 1977). "Icelandic disease (benign myalgic encephalomyelitis or Royal Free disease)". British Medical Journal. 1 (6072): 1350. doi:10.1136/bmj.1.6072.1350-a. PMC 1607215. PMID 861618.
- Straus SE (1991). "History of chronic fatigue syndrome". Reviews of Infectious Diseases. 13 Suppl 1 (Suppl. 1): S2-7. doi:10.1093/clinids/13.supplement_1.s2. PMID 2020800.
- "Press Briefing Transcripts". Centers for Disease Control and Prevention. November 3, 2006. Retrieved 2013-10-12.
- Shelokov A, Habel K, Verder E, Welsh W (August 1957). "Epidemic neuromyasthenia; an outbreak of poliomyelitislike illness in student nurses". The New England Journal of Medicine. 257 (8): 345–55. doi:10.1056/NEJM195708222570801. PMID 13464938.
- Blattner RJ (October 1956). "Benign myalgic encephalomyelitis (Akureyri disease, Iceland disease)". The Journal of Pediatrics. 49 (4): 504–6. doi:10.1016/S0022-3476(56)80241-2. PMID 13358047.
- Straus, Stephen E., ed. (1994). Chronic Fatigue Syndrome. New York, Basel, Hong Kong: Marcel Dekker Inc. p. 227. ISBN 978-0-8247-9187-2.
- Aoki T, Usuda Y, Miyakoshi H, Tamura K, Herberman RB (1987). "Low natural killer syndrome: clinical and immunologic features". Natural Immunity and Cell Growth Regulation. 6 (3): 116–28. PMID 2442602.
- Wessely S (October 1991). "History of postviral fatigue syndrome". British Medical Bulletin. 47 (4): 919–41. doi:10.1093/oxfordjournals.bmb.a072521. PMID 1794091. S2CID 12964461.
- Ramsay AM (1986). Postviral Fatigue Syndrome. The saga of Royal Free disease. London: Gower. ISBN 978-0-906923-96-2.
- Simpson LO (October 1991). "Myalgic encephalomyelitis". Journal of the Royal Society of Medicine. 84 (10): 633. PMC 1295578. PMID 1744860.
- Jason LA, Richman JA (2008). "How science can stigmatize: The case of chronic fatigue syndrome". Journal of Chronic Fatigue Syndrome. 14 (4): 85–103. doi:10.1080/10573320802092146.
- Jason LA, Holbert C, Torres-Harding S, Taylor RR (2004). "Stigma and the term chronic fatigue syndrome". Journal of Disability Policy Studies. 14 (4): 222–228. CiteSeerX 10.1.1.486.4577. doi:10.1177/10442073040140040401. S2CID 72397898.
- Evengård B, Schacterle RS, Komaroff AL (November 1999). "Chronic fatigue syndrome: new insights and old ignorance". Journal of Internal Medicine. 246 (5): 455–69. doi:10.1046/j.1365-2796.1999.00513.x. PMID 10583715. S2CID 34123925.
- Chronic fatigue syndrome; Report of a joint working group of the Royal Colleges of Physicians, Psychiatrists and General Practitioners. London: Royal Colleges of Physicians, Psychiatrists and General Practitioners. 1996. ISBN 978-1-86016-046-2.
- Tuller, David (2015-02-10). "Chronic Fatigue Syndrome Gets a New Name". well.blogs.nytimes.com. Retrieved 3 February 2021.
- "NIH PEM subgroup CDE draft recommendations" (PDF). NIH Common Data Elements project. Dec 2017.
- Reynolds KJ, Vernon SD, Bouchery E, Reeves WC (June 2004). "The economic impact of chronic fatigue syndrome". Cost Effectiveness and Resource Allocation. 2 (1): 4. doi:10.1186/1478-7547-2-4. PMC 449736. PMID 15210053.
- Jason LA, Corradi K, Torres-Harding S, Taylor RR, King C (March 2005). "Chronic fatigue syndrome: the need for subtypes". Neuropsychology Review. 15 (1): 29–58. doi:10.1007/s11065-005-3588-2. PMID 15929497. S2CID 8153255.
- Avellaneda Fernández A, Pérez Martín A, Izquierdo Martínez M, Arruti Bustillo M, Barbado Hernández FJ, de la Cruz Labrado J, Díaz-Delgado Peñas R, Gutiérrez Rivas E, Palacín Delgado C, Rivera Redondo J, Ramón Giménez JR (October 2009). "Chronic fatigue syndrome: aetiology, diagnosis and treatment". BMC Psychiatry. 9 Suppl 1: S1. doi:10.1186/1471-244X-9-S1-S1. PMC 2766938. PMID 19857242.
- Jason LA, Benton MC, Valentine L, Johnson A, Torres-Harding S (April 2008). "The economic impact of ME/CFS: individual and societal costs". Dynamic Medicine. 7: 6. doi:10.1186/1476-5918-7-6. PMC 2324078. PMID 18397528.
- Broderick G, Craddock TJ (March 2013). "Systems biology of complex symptom profiles: capturing interactivity across behavior, brain and immune regulation". Brain, Behavior, and Immunity. 29: 1–8. doi:10.1016/j.bbi.2012.09.008. PMC 3554865. PMID 23022717.
- "ME/Chronic Fatigue Syndrome Awareness Day". Centers for Disease Control and Prevention. 2017-05-12. Retrieved July 12, 2017.
- Lee, Nancy. "Dr. Nancy Lee on International Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Awareness Day". U.S. Department of Health & Human Services. Archived from the original on 2012-07-08. Retrieved 2013-10-12.
- Young DA (1995-12-23). "Florence Nightingale's fever". BMJ. 311 (7021): 1697–700. doi:10.1136/bmj.311.7021.1697. PMC 2539100. PMID 8541764.
- Feilden, Tom (2011-07-29). "'Torrent of abuse' hindering ME research". BBC News. BBC. Retrieved 2011-07-31.
- Wallace PG (October 1991). "Post-viral fatigue syndrome. Epidemiology: a critical review". British Medical Bulletin. 47 (4): 942–51. doi:10.1093/oxfordjournals.bmb.a072522. PMID 1794092.
- Mounstephen A, Sharpe M (May 1997). "Chronic fatigue syndrome and occupational health". Occupational Medicine. 47 (4): 217–27. doi:10.1093/occmed/47.4.217. PMID 9231495.
- Hooge J (1992). "Chronic fatigue syndrome: cause, controversy and care". British Journal of Nursing. 1 (9): 440–1, 443, 445–6. doi:10.12968/bjon.1992.1.9.440. PMID 1446147.
- Sharpe M (September 1996). "Chronic fatigue syndrome". The Psychiatric Clinics of North America. 19 (3): 549–73. doi:10.1016/S0193-953X(05)70305-1. PMID 8856816.
- Denz-Penhey H, Murdoch JC (April 1993). "General practitioners acceptance of the validity of chronic fatigue syndrome as a diagnosis". The New Zealand Medical Journal. 106 (953): 122–4. PMID 8474729.
- Greenlee JE, Rose JW (2000). "Controversies in neurological infectious diseases". Seminars in Neurology. 20 (3): 375–86. doi:10.1055/s-2000-9429. PMID 11051301.
- Horton-Salway M (December 2007). "The ME Bandwagon and other labels: constructing the genuine case in talk about a controversial illness" (PDF). The British Journal of Social Psychology (Submitted manuscript). 46 (Pt 4): 895–914. doi:10.1348/014466607X173456. PMID 17535450.
- Bowen J, Pheby D, Charlett A, McNulty C (August 2005). "Chronic Fatigue Syndrome: a survey of GPs' attitudes and knowledge". Family Practice. 22 (4): 389–93. doi:10.1093/fampra/cmi019. PMID 15805128.
- Larun L, Malterud K (December 2007). "Identity and coping experiences in Chronic Fatigue Syndrome: a synthesis of qualitative studies". Patient Education and Counseling. 69 (1–3): 20–8. doi:10.1016/j.pec.2007.06.008. hdl:1956/5105. PMID 17698311.
- Dumit J (February 2006). "Illnesses you have to fight to get: facts as forces in uncertain, emergent illnesses". Social Science & Medicine. 62 (3): 577–90. doi:10.1016/j.socscimed.2005.06.018. PMID 16085344.
- Lombardi VC, Ruscetti FW, Das Gupta J, Pfost MA, Hagen KS, Peterson DL, Ruscetti SK, Bagni RK, Petrow-Sadowski C, Gold B, Dean M, Silverman RH, Mikovits JA (October 2009). "Detection of an infectious retrovirus, XMRV, in blood cells of patients with chronic fatigue syndrome". Science. 326 (5952): 585–9. Bibcode:2009Sci...326..585L. doi:10.1126/science.1179052. PMC 3073172. PMID 19815723. (Retracted, see doi:10.1126/science.334.6063.1636-a)
- "No blood from chronic fatigue donors: agency". CBC. 2010-04-07. Archived from the original on April 11, 2010. Retrieved 2010-06-25.
- Atkinson, K (2010-04-21). "Chronic Fatigue Set To Disqualify Blood Donors". Voxy.co.nz. Retrieved 2010-06-25.
- "Blood Service updates CFS donor policy". Australian Red Cross Blood Service. Archived from the original on October 14, 2013. Retrieved 2013-07-07.
- "Recommendation on Chronic Fatigue Syndrome and Blood Donation". American Association of Blood Banks. 2010-06-18. Archived from the original on June 25, 2010. Retrieved 2010-06-25.
- NHS Blood and Transplant (2010-11-05). "ME/CFS sufferers permanently deferred from giving blood". Retrieved 2011-10-09.
- NHS Blood and Transplant. "Chronic Fatigue Syndrome". Retrieved 2015-02-11.
- Tuller, David (2008-05-30). "Chronic Fatigue Syndrome No Longer Seen as 'Yuppie Flu'". The New York Times. Retrieved 2015-06-29.
- Jason LA, Richman JA, Friedberg F, Wagner L, Taylor R, Jordan KM (September 1997). "Politics, science, and the emergence of a new disease. The case of chronic fatigue syndrome". The American Psychologist. 52 (9): 973–83. doi:10.1037/0003-066X.52.9.973. PMID 9301342.
- "CFS/ME – The illness and the controversy | Science Media Centre". www.sciencemediacentre.org. Science Media Centre. Archived from the original on 24 August 2018. Retrieved 26 August 2018.
- McEvedy CP, Beard AW (January 1970). "Concept of benign myalgic encephalomyelitis". British Medical Journal. 1 (5687): 11–5. doi:10.1136/bmj.1.5687.11. PMC 1700895. PMID 5411596.
- Speight, N (2013). "Myalgic encephalomyelitis/chronic fatigue syndrome: Review of history, clinical features, and controversies". Saudi Journal of Medicine & Medical Sciences. 1 (1): 11–13. doi:10.4103/1658-631x.112905.
- Cowley G, Hager M, Joseph N (1990-11-12), "Chronic Fatigue Syndrome", Newsweek: Cover Story
- Frumkin H, Packard RM, Brown P, Berkelman RL (2004). Emerging illnesses and society: negotiating the public health agenda. Baltimore: Johns Hopkins University Press. pp. 156. ISBN 978-0-8018-7942-5.
- Erlwein O, Kaye S, McClure MO, Weber J, Wills G, Collier D, Wessely S, Cleare A (January 2010). Nixon DF (ed.). "Failure to detect the novel retrovirus XMRV in chronic fatigue syndrome". PLOS ONE. 5 (1): e8519. Bibcode:2010PLoSO...5.8519E. doi:10.1371/journal.pone.0008519. PMC 2795199. PMID 20066031.
- Groom HC, Boucherit VC, Makinson K, Randal E, Baptista S, Hagan S, Gow JW, Mattes FM, Breuer J, Kerr JR, Stoye JP, Bishop KN (February 2010). "Absence of xenotropic murine leukaemia virus-related virus in UK patients with chronic fatigue syndrome". Retrovirology. 7 (1): 10. doi:10.1186/1742-4690-7-10. PMC 2839973. PMID 20156349.
- van Kuppeveld FJ, de Jong AS, Lanke KH, Verhaegh GW, Melchers WJ, Swanink CM, Bleijenberg G, Netea MG, Galama JM, van der Meer JW (February 2010). "Prevalence of xenotropic murine leukaemia virus-related virus in patients with chronic fatigue syndrome in the Netherlands: retrospective analysis of samples from an established cohort". BMJ. 340: c1018. doi:10.1136/bmj.c1018. PMC 2829122. PMID 20185493.
- Alberts B (December 2011). "Retraction". Science. 334 (6063): 1636. Bibcode:2011Sci...334.1636A. doi:10.1126/science.334.6063.1636-a. PMID 22194552.
- Lo SC, Pripuzova N, Li B, Komaroff AL, Hung GC, Wang R, Alter HJ (January 2012). "Retraction for Lo et al., Detection of MLV-related virus gene sequences in blood of patients with chronic fatigue syndrome and healthy blood donors". Proceedings of the National Academy of Sciences of the United States of America. 109 (1): 346. Bibcode:2012PNAS..109..346.. doi:10.1073/pnas.1119641109. PMC 3252929. PMID 22203980.
- "Parliamentary Briefing: Appropriate ME treatment". parliament.uk. 22 January 2019.
- "ME_Inquiry_Report" (PDF). Erythos.com. Retrieved 2011-01-28.
- "Chronic Fatigue Syndrome/Myalgic Encephalomyelitis". MRC.ac.uk. Archived from the original on January 6, 2011. Retrieved 2011-01-28.
- "APPGME.org.uk" (PDF). Retrieved 2011-01-28.
- Cohen, Jon (27 October 2015). "Criticism mounts of a long, controversial chronic fatigue study". Science. doi:10.1126/science.aad4784.
- Dove, A. (August 2000). "GAO reports on CFS funding controversy". Nat. Med. 6 (8): 846. doi:10.1038/78579. PMID 10932206. S2CID 1431198.
- "NIH takes action to bolster research on Myalgic Encephalomyelitis/Chronic Fatigue Syndrome". 29 October 2015.
- "Golden Girls, Chronic Fatigue Syndrome, and the Legacies of Hysteria". Nursing Clio. 2018-09-25. Retrieved 2019-11-14.
- Cain, Paul (2007). "Ann Bannon". Leading the parade: Conversations with America's most influential lesbians and gay men. Scarecrow Press, Inc. pp. 155–163. ISBN 0-8108-5913-0
- Hylton, Wil S. (2014-12-18). "The Unbreakable Laura Hillenbrand". The New York Times. ISSN 0362-4331. Retrieved 2020-06-27.
- Parker-Pope, Tara (2011-02-04). "An Author Escapes From Chronic Fatigue Syndrome". Well. Retrieved 2020-06-27.
- Whistler T, Unger ER, Nisenbaum R, Vernon SD (December 2003). "Integration of gene expression, clinical, and epidemiologic data to characterize Chronic Fatigue Syndrome". Journal of Translational Medicine. 1 (1): 10. doi:10.1186/1479-5876-1-10. PMC 305360. PMID 14641939.
- Kennedy G, Abbot NC, Spence V, Underwood C, Belch JJ (February 2004). "The specificity of the CDC-1994 criteria for chronic fatigue syndrome: comparison of health status in three groups of patients who fulfill the criteria". Annals of Epidemiology. 14 (2): 95–100. doi:10.1016/j.annepidem.2003.10.004. PMID 15018881.
- Aslakson E, Vollmer-Conna U, White PD (April 2006). "The validity of an empirical delineation of heterogeneity in chronic unexplained fatigue". Pharmacogenomics. 7 (3): 365–73. doi:10.2217/14622416.7.3.365. PMID 16610947.
External links
- "CDC — Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS)". Centers for Disease Control. Retrieved 2020-05-20.
| Classification | |
|---|---|
| External resources |