Sialyl-Lewis X
Sialyl LewisX (sLeX) also known as cluster of differentiation 15s (CD15s) or stage-specific embryonic antigen 1 (SSEA-1), is a tetrasaccharide carbohydrate which is usually attached to O-glycans on the surface of cells. It is known to play a vital role in cell-to-cell recognition processes. It is also the means by which an egg attracts sperm; first, to stick to it, then bond with it and eventually form a zygote. The discovery of the essential role that this tetrasaccharide plays in the fertilization process was reported in August 2011.[1]
 | |
| Names | |
|---|---|
| Other names
sialyl LeX, SLeX, CD15s, SSEA-1 | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| MeSH | sialyl+Lewis+X |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C31H52N2O23 | |
| Molar mass | 820.744 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Sialyl Lewis X is also one of the most important blood group antigens and is displayed on the terminus of glycolipids that are present on the cell surface. The sialyl Lewis X determinant, E-selectin ligand carbohydrate structure, is constitutively expressed on granulocytes and monocytes and mediates inflammatory extravasation of these cells. Resting T- and B-lymphocytes lack its expression and are induced to strongly express sialyl Lewis X upon activation. The sialyl Lewis X determinant is expressed preferentially on activated Th1 cells but not on Th2 cells.
Structure
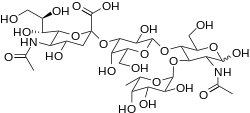
Sialyl-Lewis X is a tetrasaccharide composed of a sialic acid, fucose and an N-acetyllactosamine. Its systematic name is 5-acetylneuraminyl-(2-3)-galactosyl-(1-4)-(fucopyranosyl-(1-3))-N-acetylglucosamine (Neu5Acα2-3Galβ1-4[Fucα1-3]GlcNAcβ). In humans, according to Table 1[2] and Fig.1[3] it is synthesized by four fucosyltransferases: FUT3, FUT5, FUT6 and FUT7. The other three enzymes of the sialyltransferase family ST3GAL3, ST3GAL4,and ST3GAL6 participate in the synthesis of the Sialyl-Lewis X precursor.(Fig.1)[3]
Function
Leukocyte homing
Sialyl-Lewisx is important in leukocyte tethering and rolling. Leukocytes move through the blood stream and then tether themselves to the endothelial wall and roll along the endothelial tissue to determine if they want to leave the bloodstream to get to necessary tissue. Sialyl-Lewisx is a necessary partner for the three selectins that bind the leukocyte and endothelial cells. When sialyl-Lewisx is part of an O-glycan and attached to CD34 it can then bind to L-selectin. For the binding to L-selectin to occur sialyl-Lewisx must undergo sulfation. For sialyl-Lewisx to bind to P-selectin, an O-linked glycan near the N-terminus of P-Selectin Glycoprotein Ligand-1 (PSGL-1) is modified with sialyl-Lewisx and in combination with nearby tyrosine residues modified with sulfate, forms the binding contact for P-selectin. For sialyl-Lewisx to bind to E-selectin it can be part of either an N-linked or O-linked glycan attached to cell surface glycoproteins such as PSGL-1, CD43 or CD44. This sialyl-Lewisx mediated binding to selectins allows circulating leukocytes to stick to and roll along endothelial cells lining blood vessels thereby enabling the leukocytes to accumulate at a site of vascular inflammation.
Fertilization
Sialyl-Lewisx allows a sperm cell to recognize and fertilize an egg cell. For fertilization to occur, human sperm must bind to the zona pellucida (ZP), the translucent matrix covering the human egg composed of four glycoproteins ZP1, 2, 3, and 4, and transit through the matrix in order to fuse with the oocyte.[4] Human ZP is coated with highly dense N- and O-glycans that are terminated with the sialyl-Lewisx sequence.[1] The hemizona assay, which assesses sperm-ZP binding by counting the number of sperm bound to hemispheres of bisected nonliving human eggs in vitro, revealed that as little as 0.5 mM sialyl-Lewisx inhibits sperm-ZP binding by 63%.[1] Furthermore, adding purified and solubilized ZP3 or ZP4 from the human oocyte dose-dependently inhibits sperm-ZP binding in the hemizona assay.[5] Such evidence suggest that the early steps of human sperm-egg binding may be mediated by lectins for sialyl-Lewisx present on human sperm.
Clinical significance
Leukocyte adhesion deficiency
Defective synthesis of the sialyl Lewis X antigen results in immunodeficiency (leukocyte adhesion deficiency type 2). Defective synthesis can be caused by the loss of fucosyltransferase, impairing the glycosylation of the glycosphingolipid. Sialyl Lewis x is being researched for detection and treatment of immune disorders because of its presence on leukocytes.
Blood cancers
Sialyl-Lewisx mediates phagocytosis and chemotaxis, found on neutrophils;[6] expressed in patients with Hodgkin disease, some B-cell chronic lymphocytic leukemias, acute lymphoblastic leukemias, and most acute nonlymphocytic leukemias. CD15 is present on almost all Reed–Sternberg cells, including their rare mononuclear variants, and, as such, can be used in immunohistochemistry to identify the presence of such cells in biopsies. The presence of these cells is diagnostic of Hodgkin's lymphoma. Reed-Sternberg cells display a characteristic pattern of Sialyl-Lewisx (CD15) positivity, with membranous staining combined with staining of the Golgi apparatus. Immunohistochemical panels for the diagnosis of Hodgkins disease typically employ CD15 along with CD30 and CD45; the latter does not stain Reed-Sternberg cells, but does stain almost all other lymphoid cells. Sialyl-Lewisx is also present in about 50% of adenocarcinoma cells and can be used to distinguish such conditions from mesothelioma, which is typically negative.[7]
Cancer metastasis
Sialyl-Lewisx plays a critical role in cancer metastasis, facilitating the extravasation of cancer cells out of the bloodstream while they are moving through the body. Its expression is related to tumor stage, recurrence, and overall patient survival.[8] Therefore, sialyl Lewis x is being used as a target in studies to fight tumors and cancer cell growth. It has been shown that there is frequent overexpression of sialyl Lewis x on cancer cells and is found on both N-glycan and O-glycans. Sialyl Lewis x is being researched with CD markers to find new ways to create biosensors for cancer cells. Also, it is being used in new ways to target cancer cells specifically for cancer treatment.
In vitro fertilization
Sialyl-Lewisx is being used to achieve greater rates of fertilization of eggs in women by coating the eggs with sialyl Lewis x.
Immunity and inflammation
It plays a key role in the inflammatory response and may be used to increase the leukocyte response to infections. Sialyl Lewis x is also an inflammation-associated antigen on liver cells. It becomes over expressed on diseased liver cells and can be used as a way to detect liver disease in a patient.
Mers coronavirus binding
In June 2019, before the onset of the COVID-19 pandemic, the sulfated sialyl-Lewis X oligosaccharide (particularly with α2,3 linkages) receptor was found to be the preferred binding site, both in humans and in dromedary camels, for the coronavirus causing Middle East Respiratory Syndrome (MERS), the sixth coronavirus to be described.[9][10]
History
The term Lewis in its name comes from the name of a family of people who suffered from a red blood cell incompatibility. The studies done on these individuals' red blood cells led to the discovery of sialyl Lewis X. Sialyl Lewis x is a very important red blood cell antigen present on the glycolipids on the plasma membrane of the cell.
Its localization on the cell surface of cells led to its alternative nomenclature as a cluster of differentiation. Clusters of differentiation are a naming system devised in 1982 to classify cell-surface antigens on leukocytes identified via monoclonal antibodies. Sialyl Lewis X was assigned the name CD15.
See also
- CA19-9 (Sialyl-Lewis A)
References
- Pang, Poh-Choo; Chiu, Philip C. N.; et al. (18 August 2011). "Human Sperm Binding Is Mediated by the Sialyl-Lewisx Oligosaccharide on the Zona Pellucida". Science. 333 (6050): 1761–1764. Bibcode:2011Sci...333.1761P. doi:10.1126/science.1207438. hdl:10044/1/15584. PMID 21852454. S2CID 23610213.
- de Vries, T.; Knegtel, R. M.; Holmes, E. H.; Macher, B. A. (October 2001). "Fucosyltransferases: structure/function studies". Glycobiology. 11 (10): 119R–128R. doi:10.1093/glycob/11.10.119r. ISSN 0959-6658. PMID 11588153.
- Trinchera, Marco; Aronica, Adele; Dall’Olio, Fabio (2017-02-23). "Selectin Ligands Sialyl-Lewis a and Sialyl-Lewis x in Gastrointestinal Cancers". Biology. 6 (1): 16. doi:10.3390/biology6010016. ISSN 2079-7737. PMC 5372009. PMID 28241499.
- Clark, G. F. (2013). "The role of carbohydrate recognition during human sperm-egg binding". Human Reproduction. 28 (3): 566–577. doi:10.1093/humrep/des447. PMID 23315069.
- Chiu, P. C. N.; Wong, B. S. T.; Chung, M. -K.; Lam, K. K. W.; Pang, R. T. K.; Lee, K. -F.; Sumitro, S. B.; Gupta, S. K.; Yeung, W. S. B. (2008). "Effects of Native Human Zona Pellucida Glycoproteins 3 and 4 on Acrosome Reaction and Zona Pellucida Binding of Human Spermatozoa". Biology of Reproduction. 79 (5): 869–877. doi:10.1095/biolreprod.108.069344. PMID 18667750.
- Kerr MA, Stocks SC (November 1992). "The role of CD15-(Le(X))-related carbohydrates in neutrophil adhesion". Histochem. J. 24 (11): 811–26. doi:10.1007/BF01046353. PMID 1362195. S2CID 8602651.
- Leong, Anthony S-Y; Cooper, Kumarason; Leong, F Joel W-M (2003). Manual of Diagnostic Cytology (2nd ed.). Greenwich Medical Media, Ltd. pp. 83–84. ISBN 1-84110-100-1.
- Liang, Jin-xiao; Liang, Yong; Gao, Wei (2016-05-24). "Clinicopathological and prognostic significance of sialyl Lewis X overexpression in patients with cancer: a meta-analysis". OncoTargets and Therapy. 9: 3113–3125. doi:10.2147/OTT.S102389. ISSN 1178-6930. PMC 4888715. PMID 27307752.
- Tortorici, M. Alejandra; Walls, Alexandra C.; Lang, Yifei; Wang, Chunyan; Li, Zeshi; Koerhuis, Danielle; Boons, Geert-Jan; Bosch, Berend-Jan; Rey, Félix A.; de Groot, Raoul J.; Veesler, David (June 2019). "Structural basis for human coronavirus attachment to sialic acid receptors". Nature Structural & Molecular Biology. 26 (6): 481–489. doi:10.1038/s41594-019-0233-y. PMC 6554059. PMID 31160783.
- Li, Wentao; Hulswit, Ruben J. G.; Widjaja, Ivy; Raj, V. Stalin; McBride, Ryan; Peng, Wenjie; Widagdo, W.; Tortorici, M. Alejandra; Dieren, Brenda van; Lang, Yifei; Lent, Jan W. M. van; Paulson, James C.; Haan, Cornelis A. M. de; Groot, Raoul J. de; Kuppeveld, Frank J. M. van; Haagmans, Bart L.; Bosch, Berend-Jan (3 October 2017). "Identification of sialic acid-binding function for the Middle East respiratory syndrome coronavirus spike glycoprotein". Proceedings of the National Academy of Sciences. 114 (40): E8508–E8517. doi:10.1073/pnas.1712592114. PMC 5635925. PMID 28923942. S2CID 20912646.
Further reading
- Chen, G.-Y.; Osada, H.; Santamaria-Babi, L. F.; Kannagi, R. (7 November 2006). "Interaction of GATA-3/T-bet transcription factors regulates expression of sialyl Lewis X homing receptors on Th1/Th2 lymphocytes". Proceedings of the National Academy of Sciences. 103 (45): 16894–16899. Bibcode:2006PNAS..10316894C. doi:10.1073/pnas.0607926103. PMC 1629005. PMID 17075044.
- Etzioni, Amos; Frydman, Moshe; Pollack, Shimon; Avidor, Israeli; Phillips, M. Laurie; Paulson, James C.; Gershoni-Baruch, Ruth (17 December 1992). "Recurrent Severe Infections Caused by a Novel Leukocyte Adhesion Deficiency". New England Journal of Medicine. 327 (25): 1789–1792. doi:10.1056/NEJM199212173272505. PMID 1279426.
- Sarangapani, Krishna K.; Qian, Jin; Chen, Wei; Zarnitsyna, Veronika I.; Mehta, Padmaja; Yago, Tadayuki; McEver, Rodger P.; Zhu, Cheng (16 September 2011). "Regulation of Catch Bonds by Rate of Force Application". Journal of Biological Chemistry. 286 (37): 32749–32761. doi:10.1074/jbc.M111.240044. PMC 3173187. PMID 21775439.
- Pan, Li-Hua; Yamauchi, Kohei; Sawai, Takashi; Nakadate, Toshihide; Kojima, Yuki; Takahashi, Naofumi; Adachi, Keisuke; Kameyama, Akihiko; Inoue, Hiroshi (May 2000). "Inhibition of Binding of E- and P-selectin to Sialyl-Lewis X Molecule Suppresses the Inflammatory Response in Hypersensitivity Pneumonitis in Mice". American Journal of Respiratory and Critical Care Medicine. 161 (5): 1689–1697. doi:10.1164/ajrccm.161.5.9812016. PMID 10806176.
- Essentials of Glycobiology 3rd Edition, Chapter 14: "Structures Common to Different Glycans" https://www.ncbi.nlm.nih.gov/books/NBK453042/#_Ch14_s2_
- Stephen J Isles, Molecule of the Month: Sialyl Lewis X