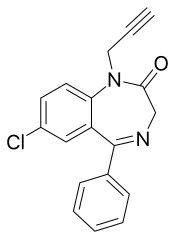
Pinazepam
Pinazepam (marketed under the brand name Domar and Duna) is a benzodiazepine drug.[1] It possesses anxiolytic, anticonvulsant, sedative and skeletal muscle relaxant properties.
 | |
 | |
| Clinical data | |
|---|---|
| Other names | 9-chloro-6-phenyl-2-prop-2-ynyl-2,5-diazabicyclo[5.4.0]undeca-5,8,10,12-tetraen-3-one |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Hepatic |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.052.650 |
| Chemical and physical data | |
| Formula | C18H13ClN2O |
| Molar mass | 308.8 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Pinazepam and its metabolite N-desmethyldiazepam are transferred to the developing fetus in utero, but the plasma drug level in the mother is usually significantly higher than in the fetus.[2]
Pinazepam differs from other benzodiazepines in that it has a propargyl group at the N-1 position of the benzodiazepine structure. It is less toxic than diazepam and in animal studies it appears to produce anxiolytic and anti-agitation properties with limited hypnotic and motor coordination impairing properties. Pinazepam is rapidly absorbed after oral administration. The main active metabolites of pinazepam are depropargylpinazepam (N-desmethyldiazepam, nordazepam) and oxazepam. In humans pinazepam acts as a pure anxiolytic agent in that it does not possess to any significant degree the other pharmacological characteristics of benzodiazepines. Its lack of intellectual, motor and hypnotic impairing effects makes it more appropriate than other benzodiazepines for day time use.[3][4][5] The elimination half-life is longer in the elderly.[6]
See also
References
- Schütz H, Holland EM, Kazemian-Erdmann F, Schölermann K (September 1988). "[Screening of the new benzodiazepine derivative, pinazepan, and its major metabolites]". Arzneimittel-Forschung. 38 (9): 1372–5. PMID 3146986.
- Pacifici GM, Cuoci L, Guarneri M, Fornaro P, Arcidiacono G, Cappelli N, et al. (1984). "Placental transfer of pinazepam and its metabolite N-desmethyldiazepam in women at term". European Journal of Clinical Pharmacology. 27 (3): 307–10. doi:10.1007/BF00542165. PMID 6150857. S2CID 1389302.
- Janbroers JM (1984). "Pinazepam: review of pharmacological properties and therapeutic efficacy". Clinical Therapeutics. 6 (4): 434–50. PMID 6147192.
- Pacifici GM, Placidi GF, Fornaro P, Gomeni R (1983). "Pharmacokinetics of pinazepam in healthy volunteers". International Journal of Clinical Pharmacology Research. 3 (5): 331–7. PMID 6147314.
- Pacifici GM, Placidi GF, Fornaro P, Gomeni R (1982). "Pinazepam: a precursor of N-desmethyldiazepam". European Journal of Clinical Pharmacology. 22 (3): 225–8. doi:10.1007/BF00545219. PMID 6809477. S2CID 1977572.
- Pacifici GM, Cuoci L, Placidi GF, Fornaro P, Gomeni R (Jan 1982). "Elimination kinetics of desmethyldiazepam in two young and two elderly subjects". European Journal of Drug Metabolism and Pharmacokinetics. 7 (1): 69–72. doi:10.1007/bf03189546. PMID 6802645. S2CID 21836038.
