Ivermectin
Ivermectin is a medication used to treat many types of parasite infestations.[3] In humans, this includes head lice, scabies, river blindness (onchocerciasis), strongyloidiasis, trichuriasis, ascariasis, and lymphatic filariasis.[3][4][5][6] In veterinary medicine, it is used to prevent and treat heartworm and acariasis, among other indications.[5] It can be taken by mouth or applied to the skin for external infestations.[3][7]
 | |
| Clinical data | |
|---|---|
| Trade names | Stromectol, Soolantra, Sklice, others |
| AHFS/Drugs.com | Systemic Monograph Topical Monograph |
| MedlinePlus | a607069 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, topical |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | – |
| Protein binding | 93% |
| Metabolism | Liver (CYP450) |
| Elimination half-life | 18 hours |
| Excretion | Feces; <1% urine |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.067.738 |
| Chemical and physical data | |
| Formula | C 48H 74O 14 (22,23-dihydroavermectin B1a) C 47H 72O 14 (22,23-dihydroavermectin B1b) |
| Molar mass | 875.106 g·mol−1 (22,23-dihydroavermectin B1a) 861.079 g·mol−1 (22,23-dihydroavermectin B1b) |
| 3D model (JSmol) | |
| |
| |
| | |
Common side effects include fever, itching, and skin rash when taken by mouth,[3] and red eyes, dry skin, and burning skin when used topically for head lice.[8] It is unclear if it is safe for use during pregnancy, but is probably acceptable for use during breastfeeding.[9] It belongs to the avermectin family of medications.[3] It works through many mechanisms of action that result in death of the targeted parasites.[3]
Ivermectin was discovered in 1975 and came into medical use in 1981.[10][11] It is on the World Health Organization's List of Essential Medicines.[12] Ivermectin is FDA-approved as an antiparasitic agent.[13]
Medical uses
Ivermectin is widely used to treat human diseases caused by roundworms and ectoparasites. For river blindness (onchocerciasis) and lymphatic filariasis, ivermectin is typically given as part of mass drug administration campaigns that distribute the drug to all members of a community affected by the disease.[14] For river blindness, a single oral dose of ivermectin (150 micrograms per kilogram of body weight) clears the body of larval Onchocerca volvulus worms for several months, preventing transmission and disease progression.[14] Adult worms survive in the skin and eventually recover to produce larval worms again. To keep the worms at bay, ivermectin is given at least once per year for the 10–15-year lifespan of the adult worms.[15] For lymphatic filariasis, oral ivermectin (200 micrograms per kilogram body weight) is part of a combination treatment given annually: ivermectin, diethylcarbamazine citrate and albendazole in places without onchocerciasis; ivermectin and albendazole in places with onchocerciasis.[16][note 1]
The World Health Organization considers ivermectin the "drug of choice" for strongyloidiasis.[18] Most cases are treated with two daily doses of oral ivermectin (200 μg per kg body weight), while severe infections are treated with five to seven days of ivermectin.[14] Ivermectin is also the primary treatment for Mansonella ozzardi and cutaneous larva migrans.[19][20] Ivermectin is sometimes added to albendazole or mebendazole for whipworm treatment, and is considered a second-line treatment for gnathostomiasis.[20][21]
Arthropod
Evidence supports its use against parasitic arthropods and insects:
- Mites such as scabies:[22][23][24] It is usually limited to cases that prove to be resistant to topical treatments or that present in an advanced state (such as Norwegian scabies).[24] One review found that the efficacy of permethrin is similar to that of systemic or topical ivermectin.[25] A separate review found that although oral ivermectin is usually effective for treatment of scabies, it does have a higher treatment failure rate than topical permethrin.[26] Another review found that oral ivermectin provided a reasonable balance between efficacy and safety.[27] Since ivermectin is more convenient than permethrin,[28] many have turned to veterinary sources of the drug to obtain assurance of a cure at an affordable price.[29]
- Lice:[30][31] Ivermectin lotion (0.5%) is FDA-approved for patients six months of age and older.[32] After a single, 10-minute application of this formulation on dry hair, 78% of subjects were found to be free of lice after two weeks.[33] This level of effectiveness is equivalent to other pediculicide treatments requiring two applications.[34]
- Bedbugs: There is tentative evidence that ivermectin kills bedbugs, as part of integrated pest management for bedbug infestations.[35][36][37] Such use however may require a prolonged course of treatment which is of unclear safety.[38]
- Malaria-bearing mosquitos, such as Anopheles gambiae: Mass drug administration of a population with ivermectin for purposes of treating/preventing nematode infestation is effective for eliminating malaria-bearing mosquitos and thereby reducing infection with residual malaria parasites.[39]
Rosacea
A review found that ivermectin was more effective than alternatives for treatment of papulopustular acne rosacea.[40][41] An ivermectin cream has been approved by the FDA, as well as in Europe, for the treatment of inflammatory lesions of rosacea. The treatment is based upon the hypothesis that parasitic mites of the genus Demodex play a role in rosacea.[42] In a clinical study, ivermectin reduced lesions by 83% over 4 months, as compared to 74% under a metronidazole standard therapy.[43]
Blepharitis
Demodex folliculorum mites have been found to be a contributing cause for blepharitis (eyelid inflammation) and a review of treatments have showed ivermectin to be an effective for treatment for reducing or eliminating mite populations and reducing symptoms.[44]
Contraindications
Ivermectin is contraindicated in children under the age of five or those who weigh less than 15 kilograms (33 pounds),[30] and individuals with liver or kidney disease.[45] Ivermectin is secreted in very low concentration in breast milk.[46] It remains unclear if ivermectin is safe during pregnancy.[47]
Adverse effects
Serious adverse events following ivermectin treatment are more common in people with very high burdens of larval Loa loa worms in their blood.[48] Those who have over 30,000 microfilaria per milliliter of blood risk inflammation and capillary blockage due to the rapid death of the microfilaria following ivermectin treatment.[48]
The main concern is neurotoxicity, which in most mammalian species may manifest as central nervous system depression, and consequent ataxia, as might be expected from potentiation of inhibitory GABA-ergic synapses.
Since drugs that inhibit the enzyme CYP3A4 often also inhibit P-glycoprotein transport, the risk of increased absorption past the blood-brain barrier exists when ivermectin is administered along with other CYP3A4 inhibitors. These drugs include statins, HIV protease inhibitors, many calcium channel blockers, lidocaine, the benzodiazepines, and glucocorticoids such as dexamethasone.[49]
For dogs, the insecticide spinosad may have the effect of increasing the toxicity of ivermectin.[50]
Pharmacology
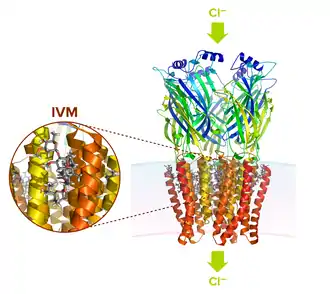
Mechanism of action
Ivermectin and its related drugs act by interfering with nerve and muscle function of helminths and insects.[51] The drug binds to glutamate-gated chloride channels that are common to invertebrate nerve and muscle cells.[52] Ivermectin binding pushes these channels open, increasing the flow of chloride ions and hyper-polarizing the cell membranes.[52][51] This hyperpolarization paralyzes the affected tissue, eventually killing the invertebrate.[52] In mammals (including humans) glutamate-gated chloride channels are restricted to the brain and spinal cord; ivermectin cannot cross the blood-brain barrier and so it does not make it to the brain to affect mammalian channels.[52]
Pharmacokinetics
Ivermectin can be given by mouth, topically, or via injection. It does not readily cross the blood–brain barrier of mammals due to the presence of P-glycoprotein,[53] (the MDR1 gene mutation affects function of this protein). Crossing may still become significant if ivermectin is given at high doses (in which case, brain levels peak 2–5 hours after administration). In contrast to mammals, ivermectin can cross the blood–brain barrier in tortoises, often with fatal consequences.
Ecotoxicity
Field studies have demonstrated the dung of animals treated with ivermectin supports a significantly reduced diversity of invertebrates, and the dung persists longer.[54]
Chemistry
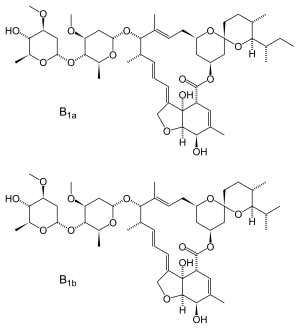
Ivermectin is an approximately 80:20 mixture of two avermectin B1 derivatives, called 22,23-dihydroavermectin B1a and B1b.[55]
History
The avermectin family of compounds was discovered by Satoshi Ōmura of Kitasato University and William Campbell of Merck. In 1970, Ōmura isolated unusual Streptomyces bacteria from the soil near a golf course along the south east coast of Honshu, Japan.[55] Ōmura sent the bacteria to William Campbell, who showed that the bacterial culture could cure mice infected with the roundworm Heligmosomoides polygyrus.[55] Campbell isolated the active compounds from the bacterial culture, naming them "avermectins" and the bacterium Streptomyces avermitilis for the compounds' ability to clear mice of worms (in Latin: a without, vermis worms).[55] Of the various avermectins, Campbell's group found the compound "avermectin B1" to be the most potent when taken orally.[55] They synthesized modified forms of avermectin B1 to improve its pharmaceutical properties, eventually choosing a mixture of at least 80% 22,23-dihydroavermectin B1a and up to 20% 22,23-dihydroavermectin B1b, a combination they called "ivermectin".[55][56]
Ivermectin was introduced in 1981.[57] Half of the 2015 Nobel Prize in Physiology or Medicine was awarded jointly to Campbell and Ōmura for discovering avermectin, "the derivatives of which have radically lowered the incidence of river blindness and lymphatic filariasis, as well as showing efficacy against an expanding number of other parasitic diseases".[58]
Society and culture
Cost
The initial price, proposed by Merck in 1987, was US$6.[59] The company donated hundreds of millions of courses of treatments since 1988 in more than 30 countries.[59] Between 1995 and 2010 the program using donated ivermectin to prevent river blindness is estimated to have prevented 7 million years of disability whilst costing US$257 million.[60]
As of 2019, the cost effectiveness of treating scabies and lice with ivermectin has not been studied.[61][62]
As of 2019, ivermectin tablets in the United States were the least expensive treatment option for lice in children at about US$10.[63] The hair lotion, however, costs about US$300 for a course of treatment.[63]
Brand names
Ivermectin is available as a generic prescription drug in the U.S. in a 3 mg tablet formulation.[64] It is also sold under the brand names Heartgard, Sklice[65] and Stromectol[66] in the United States, Ivomec worldwide by Merial Animal Health, Mectizan in Canada by Merck, Iver-DT[67] in Nepal by Alive Pharmaceutical and Ivexterm in Mexico by Valeant Pharmaceuticals International. In Southeast Asian countries, it is marketed by Delta Pharma Ltd. under the trade name Scabo 6. The formulation for rosacea treatment is sold as Soolantra. While in development, it was assigned the code MK-933 by Merck.[68]
Research
COVID-19
In vitro, ivermectin has antiviral effects against several distinct positive-sense single-strand RNA viruses, including SARS-CoV-2.[69] This has made it an attractive target for COVID-19 drug repurposing research. Subsequent studies found that ivermectin could inhibit replication of SARS-CoV-2 in monkey kidney cell culture with an IC50 of 2.2–2.8 µM.[70][71] Based on these data, however, doses much higher than the maximum approved or safely achievable for use in humans would be required for an antiviral effect.[72] Aside from practical difficulties, such high doses are not covered by current human-use approvals of the drug and would be toxic, as the antiviral mechanism of action is considered to operate via the suppression of a host cellular process,[72] specifically the inhibition of nuclear transport by importin α/β1.[73]
In November 2020, a systematic review found weak evidence of benefit when ivermectin is used as an add-on therapy for people with non-severe COVID-19.[74] A randomized controlled trial (RCT) of 24 patients with non-severe COVID-19 and no risk factors found no difference in PCR-positive nasal swabs nor in viral load between patients who received ivermectin and those given placebo, thus failing the primary outcome of the study.[75] Merck, the company from which the drug originated, has said that there is no good evidence ivermectin is plausible or effective as a drug used against COVID-19, and that attempting such use may be unsafe.[76]
As of January 2021, the U.S. National Institutes of Health COVID-19 Treatment Guidelines state that the evidence for ivermectin is too limited to allow for a recommendation for or against its use.[77] Ivermectin is not approved by the FDA for anti-viral use.[77] Additional evidence from RCTs and dose-response studies are needed.[78] At least 45 such trials were listed as of January 2021.[75]
It was reported in June 2020 that despite the absence of high-quality evidence to suggest any efficacy, use of ivermectin for prevention or treatment of early-stage COVID-19 has become increasingly widespread especially in Latin America, raising concerns about self-medication, safety, and the feasibility of future clinical trials.[79][80] In response, the Brazilian Health Regulatory Agency, Brazilian Society of Infectious Diseases, and Brazilian Thoracic Society all issued position statements in July[81] or January 2021[82][83] advising against the use of ivermectin for this purpose, and the government of Peru rescinded a previous recommendation for the use of ivermectin (alongside azithromycin and hydroxychloroquine) in hospitalized patients,[84] although as of January 2021 it is still prescribed for outpatient use.[85]Tropical diseases
Ivermectin is being studied as a potential antiviral agent against chikungunya and yellow fever.[86]
Ivermectin is also of interest in the prevention of malaria, as it is toxic to both the malaria plasmodium itself and the mosquitos that carry it.[87][88] Use of ivermectin at higher doses necessary to control malaria is probably safe, though large clinical trials have not yet been done to definitively establish the efficacy and safety of ivermectin for prophylaxis or treatment of malaria.[89]
Moxidectin has been approved by the FDA for use in people with river blindness, has a longer half-life than ivermectin, and may eventually supplant ivermectin, as it is a more potent microfilaricide, but there is a need for additional clinical trials, with long-term follow-up, to assess whether moxidectin is safe and effective for treatment of nematode infection in children and women of childbearing potential.[90][91]
NAFLD
In 2013, this antiparasitic drug was demonstrated as a novel ligand of farnesoid X receptor (FXR),[92][93] a therapeutic target for nonalcoholic fatty liver disease.[94]
Veterinary use
Ivermectin is routinely used to control parasitic worms in the gastrointestinal tract of ruminant animals. These parasites normally enter the animal when it is grazing, pass the bowel, and set and mature in the intestines, after which they produce eggs that leave the animal via its droppings and can infest new pastures. Ivermectin is effective in killing some, but not all, of these parasites.
In dogs it is routinely used as prophylaxis against heartworm.[95]
Dogs with defects in the P-glycoprotein gene (MDR1), often collie-like herding dogs, can be severely poisoned by ivermectin. The mnemonic "white feet, don't treat" refers to Scotch collies that are vulnerable to ivermectin.[96] Some other dog breeds (especially the Rough Collie, the Smooth Collie, the Shetland Sheepdog, and the Australian Shepherd), also have a high incidence of mutation within the MDR1 gene (coding for P-glycoprotein) and are sensitive to the toxic effects of ivermectin.[97][98] Clinical evidence suggests kittens are susceptible to ivermectin toxicity.[99] A 0.01% ivermectin topical preparation for treating ear mites in cats is available.[100]
Ivermectin is sometimes used as an acaricide in reptiles, both by injection and as a diluted spray. While this works well in some cases, care must be taken, as several species of reptiles are very sensitive to ivermectin. Use in turtles is particularly contraindicated.[101]
Notes
- In people with onchocerciasis, diethylcarbamazine citrate can cause a dangerous set of side effects called Mazzotti reaction. Due to this, diethylcarbamazine citrate is avoided in places where onchocerciasis is common.[17]
References
- "FDA Approves Lotion for Nonprescription Use to Treat Head Lice". U.S. Food and Drug Administration (FDA) (Press release). October 27, 2020. Retrieved October 27, 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - https://www.ema.europa.eu/documents/psusa/ivermectin-topical-use-list-nationally-authorised-medicinal-products-psusa/00010376/202004_en.pdf
- "Ivermectin". The American Society of Health-System Pharmacists. Archived from the original on January 3, 2016. Retrieved January 16, 2016.
- Sneader W (2005). Drug Discovery a History. Chichester: John Wiley & Sons. p. 333. ISBN 978-0-470-01552-0.
- Saunders Handbook of Veterinary Drugs: Small and Large Animal (4 ed.). Elsevier Health Sciences. 2015. p. 420. ISBN 978-0-323-24486-2. Archived from the original on January 31, 2016.
- CDC-Centers for Disease Control and Prevention (August 23, 2019). "Ascariasis - Resources for Health Professionals". www.cdc.gov. Retrieved December 28, 2019.
- Panahi Y, Poursaleh Z, Goldust M (2015). "The efficacy of topical and oral ivermectin in the treatment of human scabies" (PDF). Annals of Parasitology. 61 (1): 11–16. PMID 25911032.
- "Ivermectin (topical)". The American Society of Health-System Pharmacists. July 27, 2020. Retrieved January 16, 2021.
- "Ivermectin Levels and Effects while Breastfeeding". Drugs.com. Archived from the original on January 1, 2016. Retrieved January 16, 2016.
- Mehlhorn H (2008). Encyclopedia of parasitology (3rd ed.). Berlin: Springer. p. 646. ISBN 978-3-540-48994-8. Archived from the original on June 26, 2020.
- Vercruysse J, Rew RS, eds. (2002). Macrocyclic lactones in antiparasitic therapy. Oxon, UK: CABI Pub. p. Preface. ISBN 978-0-85199-840-4. Archived from the original on January 31, 2016.
- World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- Ahmed S, Karim MM, Ross AG, Hossain MS, Clemens JD, Sumiya MK, et al. (December 2020). "A five-day course of ivermectin for the treatment of COVID-19 may reduce the duration of illness". International Journal of Infectious Diseases. 103: 214–216. doi:10.1016/j.ijid.2020.11.191. PMC 7709596. PMID 33278625.
- Ashour DS (August 2019). "Ivermectin: From theory to clinical application". Int J Antimicrob Agents. 54 (2): 134–142. doi:10.1016/j.ijantimicag.2019.05.003. PMID 31071469.
- "Onchocerciasis". World Health Organization. June 14, 2019. Retrieved January 11, 2021.
- "Lymphatic filariasis". World Health Organization. March 2, 2020. Retrieved January 11, 2021.
- Babalola OE (2011). "Ocular onchocerciasis: current management and future prospects". Clin Ophthalmol. 5: 1479–91. doi:10.2147/OPTH.S8372. PMC 3206119. PMID 22069350.
- "Strongyloidiasis". World Health Organization. Retrieved January 25, 2021.
- Despommier DD, Griffin DO, Gwadz RW, Hotez PJ, Knirsch CA (2019). "26. Other Nematodes of Medical Imortance". Parasitic Diseases (PDF) (7 ed.). New York: Parasites Without Borders. p. 201. Retrieved January 26, 2021.
- Despommier DD, Griffin DO, Gwadz RW, Hotez PJ, Knirsch CA (2019). "27. Aberrant Nematode Infections". Parasitic Diseases (PDF) (7 ed.). New York: Parasites Without Borders. p. 201. Retrieved January 26, 2021.
- Despommier DD, Griffin DO, Gwadz RW, Hotez PJ, Knirsch CA (2019). "17. Trichuris trichiura". Parasitic Diseases (PDF) (7 ed.). New York: Parasites Without Borders. p. 201. Retrieved January 26, 2021.
- Brooks PA, Grace RF (August 2002). "Ivermectin is better than benzyl benzoate for childhood scabies in developing countries". Journal of Paediatrics and Child Health. 38 (4): 401–4. doi:10.1046/j.1440-1754.2002.00015.x. PMID 12174005. S2CID 22499136.
- Victoria J, Trujillo R (2001). "Topical ivermectin: a new successful treatment for scabies". Pediatric Dermatology. 18 (1): 63–5. doi:10.1046/j.1525-1470.2001.018001063.x. PMID 11207977. S2CID 39384922.
- Strong M, Johnstone P (July 2007). Strong M (ed.). "Interventions for treating scabies". The Cochrane Database of Systematic Reviews (3): CD000320. doi:10.1002/14651858.CD000320.pub2. PMC 6532717. PMID 17636630.
- Rosumeck S, Nast A, Dressler C (April 2018). "Ivermectin and permethrin for treating scabies". Cochrane Database Syst Rev. 4: CD012994. doi:10.1002/14651858.CD012994. PMC 6494415. PMID 29608022.
- Dhana A, Yen H, Okhovat JP, Cho E, Keum N, Khumalo NP (January 2018). "Ivermectin versus permethrin in the treatment of scabies: A systematic review and meta-analysis of randomized controlled trials". Journal of the American Academy of Dermatology. 78 (1): 194–198. doi:10.1016/j.jaad.2017.09.006. PMID 29241784.
- Thadanipon K, Anothaisintawee T, Rattanasiri S, Thakkinstian A, Attia J (2019). "Efficacy and safety of antiscabietic agents: A systematic review and network meta-analysis of randomized controlled trials". J Am Acad Dermatol. 80 (5): 1435–1444. doi:10.1016/j.jaad.2019.01.004. PMID 30654070.

- Crump A, Ōmura S (February 10, 2011). "Ivermectin, 'wonder drug' from Japan: the human use perspective". Proceedings of the Japan Academy. Series B, Physical and Biological Sciences. 87 (2): 13–28. Bibcode:2011PJAB...87...13C. doi:10.2183/pjab.87.13. PMC 3043740. PMID 21321478.
- Laing R, Gillan V, Devaney E (June 2017). "Ivermectin - Old Drug, New Tricks?". Trends in Parasitology. 33 (6): 463–472. doi:10.1016/j.pt.2017.02.004. PMC 5446326. PMID 28285851.

- Dourmishev AL, Dourmishev LA, Schwartz RA (December 2005). "Ivermectin: pharmacology and application in dermatology". International Journal of Dermatology. 44 (12): 981–8. doi:10.1111/j.1365-4632.2004.02253.x. PMID 16409259. S2CID 27019223.
- Strycharz JP, Yoon KS, Clark JM (January 2008). "A new ivermectin formulation topically kills permethrin-resistant human head lice (Anoplura: Pediculidae)". Journal of Medical Entomology. 45 (1): 75–81. doi:10.1603/0022-2585(2008)45[75:ANIFTK]2.0.CO;2. PMID 18283945.
- "Sklice lotion". drugs.com. Archived from the original on May 12, 2012.
- Pariser DM, Meinking TL, Bell M, Ryan WG (November 1, 2012). "Topical 0.5% ivermectin lotion for treatment of head lice". The New England Journal of Medicine. 367 (18): 1687–93. doi:10.1056/NEJMoa1200107. PMID 23113480.
- Healy M (November 1, 2012). "New treatment for lice is highly effective, study reports". Los Angeles Times. Archived from the original on January 1, 2020.
- Crump A (May 2017). "Ivermectin: enigmatic multifaceted 'wonder' drug continues to surprise and exceed expectations". J. Antibiot. 70 (5): 495–505. doi:10.1038/ja.2017.11. PMID 28196978.
- Ōmura S (August 2016). "A Splendid Gift from the Earth: The Origins and Impact of the Avermectins (Nobel Lecture)". Angew. Chem. Int. Ed. Engl. 55 (35): 10190–209. doi:10.1002/anie.201602164. PMID 27435664.
- James WD, Elston D, Berger T, Neuhaus I (2015). Andrews' Diseases of the Skin: Clinical Dermatology. Elsevier Health Sciences. p. 439. ISBN 9780323319690.
Ivermectin treatment is emerging as a potential ancillary measure.
- Lebwohl MG, Heymann WR, Berth-Jones J, Coulson I (2017). Treatment of Skin Disease: Comprehensive Therapeutic Strategies. Elsevier Health Sciences. p. 89. ISBN 9780702069130.
- Tizifa TA, Kabaghe AN, McCann RS, van den Berg H, Van Vugt M, Phiri KS (2018). "Prevention Efforts for Malaria". Curr Trop Med Rep. 5 (1): 41–50. doi:10.1007/s40475-018-0133-y. PMC 5879044. PMID 29629252.
- Husein-ElAhmed H, Steinhoff M (January 2020). "Efficacy of topical ivermectin and impact on quality of life in patients with papulopustular rosacea: A systematic review and meta-analysis". Dermatol Ther. 33 (1): e13203. doi:10.1111/dth.13203. PMID 31863543. S2CID 209433363.
- Siddiqui K, Stein Gold L, Gill J (2016). "The efficacy, safety, and tolerability of ivermectin compared with current topical treatments for the inflammatory lesions of rosacea: a network meta-analysis". SpringerPlus. 5 (1): 1151. doi:10.1186/s40064-016-2819-8. PMC 4956638. PMID 27504249.
- Moran EM, Foley R, Powell FC (2017). "Demodex and rosacea revisited". Clin. Dermatol. 35 (2): 195–200. doi:10.1016/j.clindermatol.2016.10.014. PMID 28274359.
- "Galderma Receives FDA Approval of Soolantra (Ivermectin) Cream for Rosacea". drugs.com. Archived from the original on January 22, 2015."
- Navel V, Mulliez A, Benoist d'Azy C, Baker JS, Malecaze J, Chiambaretta F, Dutheil F (October 2019). "Efficacy of treatments for Demodex blepharitis: A systematic review and meta-analysis". The Ocular Surface. 17 (4): 655–669. doi:10.1016/j.jtos.2019.06.004. PMID 31229586.
- Heukelbach J, Winter B, Wilcke T, Muehlen M, Albrecht S, de Oliveira FA, Kerr-Pontes LR, Liesenfeld O, Feldmeier H (August 2004). "Selective mass treatment with ivermectin to control intestinal helminthiases and parasitic skin diseases in a severely affected population". Bulletin of the World Health Organization. 82 (8): 563–71. PMC 2622929. PMID 15375445.
- Koh YP, Tian EA, Oon HH (September 2019). "New changes in pregnancy and lactation labelling: Review of dermatologic drugs". Int J Womens Dermatol. 5 (4): 216–226. doi:10.1016/j.ijwd.2019.05.002. PMC 6831768. PMID 31700976.
- Nicolas P, Maia MF, Bassat Q, Kobylinski KC, Monteiro W, Rabinovich NR, Menéndez C, Bardají A, Chaccour C (January 2020). "Safety of oral ivermectin during pregnancy: a systematic review and meta-analysis". Lancet Glob Health. 8 (1): e92–e100. doi:10.1016/S2214-109X(19)30453-X. PMID 31839144.
- Pion SD, Tchatchueng-Mbougua JB, Chesnais CB, Kamgno J, Gardon J, Chippaux JP, et al. (April 2019). "Effect of a Single Standard Dose (150–200 μg/kg) of Ivermectin on Loa loa Microfilaremia: Systematic Review and Meta-analysis". Open Forum Infectious Diseases. 6 (4): ofz019. doi:10.1093/ofid/ofz019. PMC 6449757. PMID 30968052.
- Goodman and Gilman's The Pharmacological Basis of Therapeutics], 11th edition, pages 1084–1087.
- "COMFORTIS® and ivermectin interaction Safety Warning Notification". U.S. Food and Drug Administration (FDA) Center for Veterinary Medicine (CVM). Archived from the original on August 29, 2009.
- Martin RJ, Robertson AP, Choudhary S (January 2021). "Ivermectin: An Anthelmintic, an Insecticide, and Much More". Trends Parasitol. 37 (1): 48–64. doi:10.1016/j.pt.2020.10.005. PMID 33189582. S2CID 226972704.
- Omura S, Crump A (September 2014). "Ivermectin: panacea for resource-poor communities?". Trends Parasitol. 30 (9): 445–55. doi:10.1016/j.pt.2014.07.005. PMID 25130507.
- Borst P, Schinkel AH (June 1996). "What have we learnt thus far from mice with disrupted P-glycoprotein genes?". European Journal of Cancer. 32A (6): 985–90. doi:10.1016/0959-8049(96)00063-9. PMID 8763339.
- Iglesias LE, Saumell CA, Fernández AS, Fusé LA, Lifschitz AL, Rodríguez EM, Steffan PE, Fiel CA (December 2006). "Environmental impact of ivermectin excreted by cattle treated in autumn on dung fauna and degradation of faeces on pasture". Parasitology Research. 100 (1): 93–102. doi:10.1007/s00436-006-0240-x. PMID 16821034. S2CID 28765870.
- Laing R, Gillan V, Devaney E (June 2017). "Ivermectin - Old Drug, New Tricks?". Trends Parasitol. 33 (6): 463–472. doi:10.1016/j.pt.2017.02.004. PMC 5446326. PMID 28285851.
- Campbell WC, Fisher MH, Stapley EO, Albers-Schönberg G, Jacob TA (August 1983). "Ivermectin: a potent new antiparasitic agent". Science. 221 (4613): 823–8. Bibcode:1983Sci...221..823C. doi:10.1126/science.6308762. PMID 6308762.
- Campbell WC, Burg RW, Fisher MH, Dybas RA (June 26, 1984). "Chapter 1: The discovery of ivermectin and other avermectins". Pesticide Synthesis Through Rational Approaches. ACS Symposium Series. 255. American Chemical Society. pp. 5–20. doi:10.1021/bk-1984-0255.ch001. ISBN 978-0-8412-1083-7.
- "The Nobel Prize in Physiology or Medicine 2015" (PDF). Nobel Foundation. Archived from the original (PDF) on October 6, 2015. Retrieved October 7, 2015.
- Crump A, Ōmura S (2011). "Ivermectin, 'wonder drug' from Japan: the human use perspective". Proceedings of the Japan Academy. Series B, Physical and Biological Sciences. 87 (2): 13–28. Bibcode:2011PJAB...87...13C. doi:10.2183/pjab.87.13. PMC 3043740. PMID 21321478.
- Omaswa F, Crisp N (2014). African Health Leaders: Making Change and Claiming the Future. OUP Oxford. p. PT158. ISBN 9780191008412.
- Chiu S, Argaez C (2019). Ivermectin for Parasitic Skin Infections of Scabies: A Review of Comparative Clinical Effectiveness, Cost-Effectiveness, and Guidelines. CADTH Rapid Response Reports. Ottawa (ON): Canadian Agency for Drugs and Technologies in Health. PMID 31424718.
- Young C, Argáez C (2019). Ivermectin for Parasitic Skin Infections of Lice: A Review of Comparative Clinical Effectiveness, Cost-Effectiveness, and Guidelines. CADTH Rapid Response Reports. Ottawa (ON): Canadian Agency for Drugs and Technologies in Health. PMID 31487135.
- Kliegman RM, St Geme J (2019). Nelson Textbook of Pediatrics E-Book. Elsevier Health Sciences. p. 3575. ISBN 9780323568883.
- U.S. FDA. "Abbreviated New Drug Application (ANDA): 204154". Drugs@FDA: FDA Approved Drug Products. U.S. Food and Drug Administration. Retrieved August 18, 2018.
- "Sklice- ivermectin lotion". DailyMed. November 9, 2017. Retrieved October 28, 2020.
- "Stromectol- ivermectin tablet". DailyMed. December 15, 2019. Retrieved October 28, 2020.
- Adhikari S (May 27, 2014). "Alive Pharmaceutical (P) LTD.: Iver-DT". Alive Pharmaceutical (P) LTD. Archived from the original on March 4, 2016. Retrieved October 7, 2015.
- Pampiglione S, Majori G, Petrangeli G, Romi R (1985). "Avermectins, MK-933 and MK-936, for mosquito control". Transactions of the Royal Society of Tropical Medicine and Hygiene. 79 (6): 797–9. doi:10.1016/0035-9203(85)90121-X. PMID 3832491.
- Heidary F, Gharebaghi R (September 2020). "Ivermectin: a systematic review from antiviral effects to COVID-19 complementary regimen". The Journal of Antibiotics. 73 (9): 593–602. doi:10.1038/s41429-020-0336-z. PMC 7290143. PMID 32533071.
- Caly L, Druce JD, Catton MG, Jans DA, Wagstaff KM (June 2020). "The FDA-approved drug ivermectin inhibits the replication of SARS-CoV-2 in vitro". Antiviral Research. 178: 104787. doi:10.1016/j.antiviral.2020.104787. PMC 7129059. PMID 32251768.

- Şimşek Yavuz S, Ünal S (April 2020). "Antiviral treatment of COVID-19". Turkish Journal of Medical Sciences. 50 (SI-1): 611–619. doi:10.3906/sag-2004-145. PMC 7195979. PMID 32293834.
- Bray M, Rayner C, Noël F, Jans D, Wagstaff K (June 2020). "Ivermectin and COVID-19: A report in Antiviral Research, widespread interest, an FDA warning, two letters to the editor and the authors' responses". Antiviral Research. 178: 104805. doi:10.1016/j.antiviral.2020.104805. PMC 7172803. PMID 32330482.
- Yang SN, Atkinson SC, Wang C, Lee A, Bogoyevitch MA, Borg NA, Jans DA (May 2020). "The broad spectrum antiviral ivermectin targets the host nuclear transport importin α/β1 heterodimer" (PDF). Antiviral Research. 177: 104760. doi:10.1016/j.antiviral.2020.104760. PMID 32135219.
- Padhy BM, Mohanty RR, Das S, Meher BR (2020). "Therapeutic potential of ivermectin as add on treatment in COVID 19: A systematic review and meta-analysis". Journal of Pharmacy & Pharmaceutical Sciences. 23: 462–469. doi:10.18433/jpps31457. PMID 33227231.
the complication rate and mortality amongst patients with severe disease have been reported to be very high. In such patients the effectiveness of add on ivermectin has not yet been explored
- Chaccour C, Casellas A, Blanco-Di Matteo A, Pineda I, Fernandez-Montero A, Ruiz-Castillo P, et al. (January 2021). "The effect of early treatment with ivermectin on viral load, symptoms and humoral response in patients with non-severe COVID-19: A pilot, double-blind, placebo-controlled, randomized clinical trial". EClinicalMedicine. Elsevier BV: 100720. doi:10.1016/j.eclinm.2020.100720. PMID 33495752.
- "Merck Statement on Ivermectin use During the COVID-19 Pandemic". Merck. February 4, 2021.
- "The COVID-19 Treatment Guidelines Panel's Statement on the Use of Ivermectin for the Treatment of COVID-19". National Institutes of Health. January 14, 2020.
- Kaur H, Shekhar N, Sharma S, Sarma P, Prakash A, Medhi B (January 2021). "Ivermectin as a potential drug for treatment of COVID-19: an in-sync review with clinical and computational attributes". Pharmacological Reports. doi:10.1007/s43440-020-00195-y. PMC 7778723. PMID 33389725.
- Mega ER (October 2020). "Latin America's embrace of an unproven COVID treatment is hindering drug trials". Nature. 586 (7830): 481–482. doi:10.1038/d41586-020-02958-2. PMID 33077974.
- Molento MB (December 2020). "COVID-19 and the rush for self-medication and self-dosing with ivermectin: A word of caution". One Health. Elsevier BV. 10: 100148. doi:10.1016/j.onehlt.2020.100148. PMID 32632377.
- Quintanilha, Dayana de Oliveira (July 13, 2020). "Anvisa se manifesta contra o uso da ivermectina na Covid-19" [Anvisa speaks out against the use of ivermectin in Covid-19]. PEBMED (in Portuguese).
- "Atualizações e Recomendações sobre a Covid-19" [Updates and recommendations on Covid-19] (PDF) (in Portuguese). Sociedade Brasileira de Infectologia. December 9, 2020.
- "Posicionamento da Sociedade Brasileira de Pneumologia e Tisiologia Sobre o Colapso em Manaus e Tratamento Preventivo e Precoce da Covid-19" [Brazilian Thoracic Society Position Statement on the Collapse in Manaus and the Preventive and Early Treatment of Covid-19] (in Portuguese). Sociedade Brasileira de Pneumologia e Tisiologia. January 17, 2021. Retrieved January 18, 2021.
- Andina (October 13, 2020). "Covid-19: Minsa aprueba resolución que deja sin efecto el uso de tres medicamentos" (in Spanish). Agencia Peruana de Noticias.
- "Ivermectina vuelve a ser incluida en tratamiento covid-19" (in Spanish). Extra. January 22, 2021.
- Varghese FS, Kaukinen P, Gläsker S, Bespalov M, Hanski L, Wennerberg K, Kümmerer BM, Ahola T (February 2016). "Discovery of berberine, abamectin and ivermectin as antivirals against chikungunya and other alphaviruses". Antiviral Research. 126: 117–24. doi:10.1016/j.antiviral.2015.12.012. PMID 26752081.
- Chaccour C, Hammann F, Rabinovich NR (April 2017). "Ivermectin to reduce malaria transmission I. Pharmacokinetic and pharmacodynamic considerations regarding efficacy and safety". Malaria Journal. 16 (1): 161. doi:10.1186/s12936-017-1801-4. PMC 5402169. PMID 28434401.
- Siewe Fodjo JN, Kugler M, Hotterbeekx A, Hendy A, Van Geertruyden JP, Colebunders R (August 2019). "Would ivermectin for malaria control be beneficial in onchocerciasis-endemic regions?". Infectious Diseases of Poverty. 8 (1): 77. doi:10.1186/s40249-019-0588-7. PMC 6706915. PMID 31439040.
- Navarro M, Camprubí D, Requena-Méndez A, Buonfrate D, Giorli G, Kamgno J, Gardon J, Boussinesq M, Muñoz J, Krolewiecki A (April 2020). "Safety of high-dose ivermectin: a systematic review and meta-analysis". J. Antimicrob. Chemother. 75 (4): 827–834. doi:10.1093/jac/dkz524. PMID 31960060.
- Maheu-Giroux M, Joseph SA (August 2018). "Moxidectin for deworming: from trials to implementation". The Lancet. Infectious Diseases. 18 (8): 817–819. doi:10.1016/S1473-3099(18)30270-6. PMID 29858152.
- Boussinesq M (October 2018). "A new powerful drug to combat river blindness". Lancet. 392 (10154): 1170–1172. doi:10.1016/S0140-6736(18)30101-6. PMID 29361336.

- Carotti A, Marinozzi M, Custodi C, Cerra B, Pellicciari R, Gioiello A, Macchiarulo A (2014). "Beyond bile acids: targeting Farnesoid X Receptor (FXR) with natural and synthetic ligands". Current Topics in Medicinal Chemistry. 14 (19): 2129–42. doi:10.2174/1568026614666141112094058. PMID 25388537.
- Jin L, Feng X, Rong H, Pan Z, Inaba Y, Qiu L, et al. (2013). "The antiparasitic drug ivermectin is a novel FXR ligand that regulates metabolism". Nature Communications. 4: 1937. Bibcode:2013NatCo...4.1937J. doi:10.1038/ncomms2924. PMID 23728580.
- Kim SG, Kim BK, Kim K, Fang S (December 2016). "Bile Acid Nuclear Receptor Farnesoid X Receptor: Therapeutic Target for Nonalcoholic Fatty Liver Disease". Endocrinology and Metabolism. 31 (4): 500–504. doi:10.3803/EnM.2016.31.4.500. PMC 5195824. PMID 28029021.
- Papich MG (January 1, 2016). "Ivermectin". In Papich MG (ed.). Saunders Handbook of Veterinary Drugs. Saunders Handbook of Veterinary Drugs (Fourth Edition). W.B. Saunders. pp. 420–423. doi:10.1016/B978-0-323-24485-5.00323-5. ISBN 978-0-323-24485-5. Retrieved April 7, 2020.
- Dowling P (December 2006). "Pharmacogenetics: it's not just about ivermectin in collies". Can. Vet. J. 47 (12): 1165–8. PMC 1636591. PMID 17217086.
- "MDR1 FAQs". Australian Shepherd Health & Genetics Institute, Inc. Archived from the original on December 13, 2007.
- "Multidrug Sensitivity in Dogs". Washington State University's College of Veterinary Medicine. Archived from the original on June 23, 2015.
- Frischke H, Hunt L (April 1991). "Alberta. Suspected ivermectin toxicity in kittens". The Canadian Veterinary Journal. 32 (4): 245. PMC 1481314. PMID 17423775.
- "Acarexx". Boehringer Ingelheim. April 11, 2016.
- Klingenberg R (2007). Understanding reptile parasites: from the experts at Advanced Vivarium Systems. Irvine, Calif: Advanced Vivarium Systems. ISBN 978-1882770908.
External links
- "Ivermectin". Drug Information Portal. U.S. National Library of Medicine.
- The Carter Center River Blindness (Onchocerciasis) Control Program
- "ivermectin (Rx) Stromectol". Medscape.
- "Ivermectin Topical". MedlinePlus.