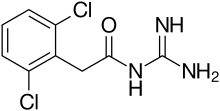
Guanfacine
Guanfacine, sold under the brand name Tenex among others, is an oral medication used to treat attention deficit hyperactivity disorder (ADHD) and high blood pressure.[5] It is not considered a first-line treatment for either indication.[5]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Estulic, Intuniv, Tenex, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601059 |
| License data |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 80-100% (IR), 58% (XR)[1][2] |
| Protein binding | 70%[1][2] |
| Metabolism | CYP3A4[1][2] |
| Elimination half-life | IR: 10-17 hours; XR: 17 hours (10-30) in adults & adolescents and 14 hours in children[1][2][3][4] |
| Excretion | Kidney (80%; 50% [range: 40-75%] as unchanged drug)[1][2] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.044.933 |
| Chemical and physical data | |
| Formula | C9H9Cl2N3O |
| Molar mass | 246.09 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Common side effects include sleepiness, constipation, dry mouth, sexual problems, and headaches.[5] Other side effect may include anxiety, low blood pressure, depression, and urinary problems.[6] Use is not recommended during pregnancy or breastfeeding.[6] It appears to work by activating the α2A receptors in the brain thereby decreasing sympathetic nervous system activity.[5]
Guanfacine was approved for medical use in the United States in 1986.[5] It is available as a generic medication.[5] In 2017, it was the 131st most commonly prescribed medication in the United States, with more than five million prescriptions.[7][8]
Medical uses

Guanfacine is used alone or with stimulants to treat people with attention deficit hyperactivity disorder.[9][10] It is also used to treat high blood pressure.[2] It is a less preferred treatment for ADHD (after more established treatments such as methylphenidate and amphetamines) and for high blood pressure.[5]
Adverse effects
Side effects of guanfacine are dose-dependent.[11]
Very common (>10% incidence) adverse effects include sleepiness, tiredness, headache, and stomach ache.[12]
Common (1-10% incidence) adverse effects include decreased appetite, depressed mood, anxiety, irritability, mood changes, insomnia, nightmares, dizziness, lack of energy, slowed heart beat, low blood pressure, feeling faint when standing quickly, vomiting, nausea, diarrhea, constipation, dry mouth, urinary incontinence, and rashes.[12]
Typical side effects such as fatigue, irritability and stomach upset can take a week or two to subside. Increases in dosage can have the same adjustment period.
Interactions
Guanfacine availability is significantly affected by the CYP3A4 and CYP3A5 enzymes. Medications that inhibit or induce those enzymes change the amount of guanfacine in circulation and thus its efficacy and rate of adverse effects; likewise, guanfacine affects those medications. Because of its impact on the heart, it should be used with caution with other cardioactive drugs. Similar concern is appropriate when it is used with sedating medications.[12]
Guanfacine is known to lower the user's tolerance for alcohol, heightening its effect, and alcohol use may prolong the effects of the medication.[13]
Pharmacology
Guanfacine is a highly selective agonist of the α2A adrenergic receptor, with low affinity for other receptors.[14] However it may also be a potent 5-HT2B receptor agonist, potentially contributing to valvulopathy.[15]
Mechanism of action
Guanfacine works by activating α2A adrenoceptors in the central nervous system. This results in reduced peripheral sympathetic outflow and thus a reduction in peripheral sympathetic tone, which lowers both systolic and diastolic blood pressure.[16] In ADHD, guanfacine works by strengthening regulation of attention and behavior by the prefrontal cortex.[17] These enhancing effects on prefrontal cortical functions are thought to be due to drug stimulation of post-synaptic α2A adrenoceptors on dendritic spines. cAMP-mediated opening of HCN and KCNQ channels is inhibited and thus prefrontal cortical synaptic connectivity and neuronal firing are enhanced.[17][18] The use of guanfacine for treating prefrontal disorders was developed by the Arnsten Lab at Yale University based on understanding the needs of the prefrontal cortex.[17][19]
Pharmacokinetics
Guanfacine has an oral bioavailability of 80%. There is no clear evidence of any first-pass metabolism. Elimination half-life is 17 hours with the major elimination route being renal. The principal metabolite is the 3-hydroxy-derivative, with evidence of moderate biotransformation, and the key intermediate is an epoxide.[20] Elimination is not impacted with impaired renal function. As such, metabolism by liver is the assumption for those with impaired renal function, as supported by increased frequency of known side effects of orthostatic hypotension and sedation.[21]
Drug Synthesis
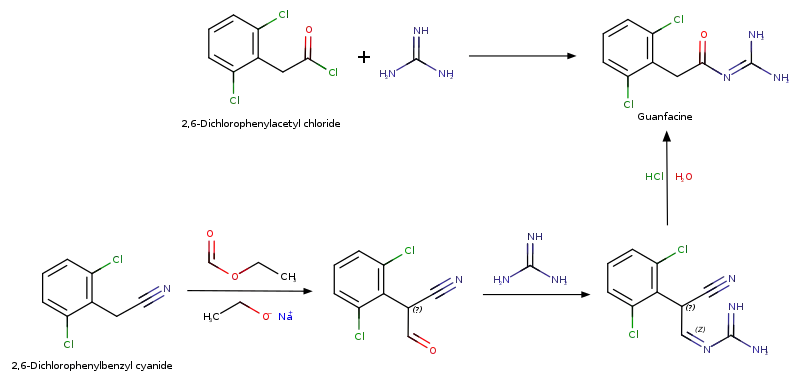
History
In 1986, guanfacine was approved by the FDA for the treatment of hypertension under the brand name Tenex (Drugs@FDA). In 2010, guanfacine was approved by the FDA for the treatment of attention deficit hyperactivity disorder for people 6–17 years old.[9] It was approved for ADHD by the European Medicines Agency under the name Intuniv in 2015.[22] It was added to the Australian Pharmaceutical Benefits Scheme for the treatment of ADHD in 2018.[23]
Brand names
Brand names include Tenex, Afken, Estulic, and Intuniv (an extended release formulation).
Research
Guanfacine has been studied as a treatment for post-traumatic stress disorder (PTSD). Evidence of efficacy in adults is limited but one study found positive results in children with comorbid ADHD.[24] It may be also useful in adult PTSD patients who do not respond to SSRIs.[25]
Results of studies using guanfacine to treat Tourette's have been mixed.[26]
Guanfacine has been investigated for treatment of withdrawal for opioids, ethanol, and nicotine.[27] Guanfacine has been shown to help reduce stress-induced craving of nicotine in smokers trying to quit, which may involve strengthening of prefrontal cortical self-control.[28]
References
- "Guanfacine (guanfacine) Tablet [Genpharm Inc.]". DailyMed. Genpharm Inc. March 2007. Retrieved 9 November 2013.
- "guanfacine (Rx) - Intuniv, Tenex". Medscape Reference. WebMD. Retrieved 9 November 2013.
- Hofer, Kristi N.; Buck, Marcia L. (2008). "New Treatment Options for Attention-Deficit/Hyperactivity Disorder (ADHD): Part II. Guanfacine". Pediatric Pharmacotherapy (14): 4.
- Cruz, MP (Aug 2010). "Guanfacine Extended-Release Tablets (Intuniv), a Nonstimulant Selective Alpha(2A)-Adrenergic Receptor Agonist For Attention-Deficit/Hyperactivity Disorder". Pharmacy and Therapeutics. 35 (8): 448–51. PMC 2935643. PMID 20844694.
- "Guanfacine Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Archived from the original on 4 January 2018. Retrieved 18 March 2019.
- British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. pp. 349–350. ISBN 9780857113382.
- "The Top 300 of 2020". ClinCalc. Retrieved 11 April 2020.
- "Guanfacine - Drug Usage Statistics". ClinCalc. Retrieved 11 April 2020.
- Kornfield R, Watson S, Higashi A, Dusetzina S, Conti R, Garfield R, Dorsey ER, Huskamp HA, Alexander GC (April 2013). "Impact of FDA Advisories on Pharmacologic Treatment of Attention Deficit Hyperactivity Disorder". Psychiatric Services. 64 (4): 339–46. doi:10.1176/appi.ps.201200147. PMC 4023684. PMID 23318985.
- Zito, Julie M.; Derivan, Albert T.; Kratochvil, Christopher J.; Safer, Daniel J.; Fegert, Joerg M.; Greenhill, Laurence L. (15 September 2008). "Off-label psychopharmacologic prescribing for children: History supports close clinical monitoring". Child and Adolescent Psychiatry and Mental Health. 2 (1): 24. doi:10.1186/1753-2000-2-24. PMC 2566553. PMID 18793403.

- Jerie, P. (1980). "Clinical experience with guanfacine in long-term treatment of hypertension: Part II: adverse reactions to guanfacine". British Journal of Clinical Pharmacology. 10 (Suppl 1): 157S–164S. doi:10.1111/j.1365-2125.1980.tb04924.x. PMC 1430125. PMID 6994770.
- "Intuniv 1 mg, 2 mg, 3 mg, 4 mg prolonged-release tablets - Summary of Product Characteristics". UK Electronic Medicines Compendium. June 2017.
- "Guanfacine | Side Effects, Dosage, Uses, and More".
- Roth, BL; Driscol, J (12 January 2011), "PDSP Ki Database", Psychoactive Drug Screening Program (PDSP), University of North Carolina at Chapel Hill and the United States National Institute of Mental Health, archived from the original on 8 November 2013, retrieved 15 November 2013
- Huang, Xi-Ping; Setola, Vincent; Yadav, Prem N.; Allen, John A.; Rogan, Sarah C.; Hanson, Bonnie J.; Revankar, Chetana; Robers, Matt; Doucette, Chris; Roth, Bryan L. (2009). "Parallel Functional Activity Profiling Reveals Valvulopathogens Are Potent 5-Hydroxytryptamine2B Receptor Agonists: Implications for Drug Safety Assessment". Molecular Pharmacology. 76 (4): 710–22. doi:10.1124/mol.109.058057. PMC 2769050. PMID 19570945.
- Van Zwieten, P.; Thoolen, M. & Timmermans, P. (1983). "The pharmacology of centrally acting antihypertensive drugs". British Journal of Clinical Pharmacology. 15 (Suppl 4): 455S–462S. doi:10.1111/j.1365-2125.1983.tb00311.x. PMC 1427667.
- Arnsten AF (October 2010), "The use of α2A adrenergic agonists for the treatment of attention-deficit/hyperactivity disorder", Expert Review of Neurotherapeutics, 10 (10): 1595–605, doi:10.1586/ern.10.133, PMC 3143019, PMID 20925474
- Wang, m; et al. (2007). "Alpha2A-adrenoceptors strengthen working memory networks by inhibiting cAMP-HCN channel signaling in prefrontal cortex". Cell. 129 (2): 397–410. doi:10.1016/j.cell.2007.03.015. PMID 17448997. S2CID 741677.
- Arnsten&Jin (2012). "Guanfacine for the treatment of cognitive disorders: a century of discoveries at Yale". Yale J Biol Med. 85 (1): 45–58. PMC 3313539. PMID 22461743.
- Kiechel, J. (1980). "Pharmacokinetics and metabolism of guanfacine in man: A review". British Journal of Clinical Pharmacology. 10 (Suppl 1): 25S–32S. doi:10.1111/j.1365-2125.1980.tb04901.x. PMC 1430131. PMID 6994775.
- Kirch, W.; Kohler, H. & Braun, W. (1980). "Elimination of guanfacine in patients with normal and impaired renal function". British Journal of Clinical Pharmacology. 10 (Suppl 1): 33S–35S. doi:10.1111/j.1365-2125.1980.tb04902.x. PMC 1430110. PMID 6994776.
- "European Medicines Agency: Intuniv". ema.europa.eu. October 2015.
- "New drugs listed on the PBS for rheumatoid arthritis, cystic fibrosis and ADHD". Royal Australian College of General Practitioners. Retrieved 11 September 2018.
- Connor, Daniel F.; Grasso, Damion J.; Slivinsky, Michelle D.; Pearson, Geraldine S.; Banga, Alok (May 2013). "An Open-Label Study of Guanfacine Extended Release for Traumatic Stress Related Symptoms in Children and Adolescents". Journal of Child and Adolescent Psychopharmacology. 23 (4): 244–251. doi:10.1089/cap.2012.0119. ISSN 1044-5463. PMC 3657282. PMID 23683139.
- Belkin, MR; Schwartz, TL (2015). "Alpha-2 receptor agonists for the treatment of posttraumatic stress disorder". Drugs in Context. 4: 212286. doi:10.7573/dic.212286. PMC 4544272. PMID 26322115.
- Srour M, Lespérance P, Richer F, Chouinard S (2008). "Psychopharmacology of Tic Disorders". J Can Acad Child Adolesc Psychiatry. 17 (3): 150–159. PMC 2527768. PMID 18769586.
- Sofuogul, M. & Sewell, A. (2009), "Norepinephrine and Stimulant Addiction", Addiction Biology, 14 (2): 119–129, doi:10.1111/j.1369-1600.2008.00138.x, PMC 2657197, PMID 18811678
- McKee, SA; Potenza, MN; Kober, H; Sofuoglu, M; Arnsten, AF; Picciotto, MR; Weinberger, AH; Ashare, R; Sinha, R (Mar 2015). "A translational investigation targeting stress-reactivity and prefrontal cognitive control with guanfacine for smoking cessation". J. Psychopharmacol. 29 (3): 300–311. doi:10.1177/0269881114562091. PMC 4376109. PMID 25516371.
External links
- "Guanfacine". Drug Information Portal. U.S. National Library of Medicine.