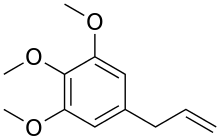
Elemicin
Elemicin is a phenylpropene, a natural organic compound, and is a constituent of several plant species' essential oils.[1][2]
 | |
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.006.954 |
| Chemical and physical data | |
| Formula | C12H16O3 |
| Molar mass | 208.257 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Natural occurrence
Elemicin is a constituent of the oleoresin and the essential oil of Canarium luzonicum (also referred to as elemi). Elemicin is named after this tree. One study found it to compose 2.4% of the fresh essential oil.[1] Elemicin is also present in the oils of the spices nutmeg and mace, with it composing 2.4% and 10.5% of those oils respectively.[2] Structurally, elemcin is similar to myristicin, differing only by myristicin's methyl group that joins the two oxygen atoms that make up its dioxymethy moiety, with both constituents being found in nutmeg and mace.
Isolation
Elemicin was first isolated from elemi oil using vacuum distillation. Specifically, the substance was collected between 162-165 °C at a reduced pressure of 10 torr.[3][4]
Preparation
Elemicin has been synthesized from syringol and allyl bromide using Williamson ether synthesis and Claisen rearrangement.[5][6] The electrophilic aromatic substitution entering the para-position was made possible by secondary Cope rearrangement.[7] This is due to syringol's allyl aromatic ether being blocked by ethers in both ortho-positions. When blocked the allyl group migrates to the para-position, in this case with yields above 85%.[8]
Pharmacology
Raw nutmeg causes anticholinergic-like effects, which are attributed to elemicin and myristicin.[10][11]
See also
References
- Villanueva MA, Torres RC, Baser KH, Özek T, Kürkçüoglu M (1993). "The Composition of Manila Elemi Oil". Flavour and Fragrance Journal. 8: 35–37. doi:10.1002/ffj.2730080107.
- Leela N (2008). Chemistry of Spices. Calicut, Kerala, India: Biddles Ltd. pp. 165–188 [170]. ISBN 9781845934057.
- "Constituents of Essential Oils. Elemicin, a High-boiling Constituent of Elemi Oil, and the Displacement of Alkyloxygroups in the Benzene Nucleus by Hydrogen". Journal of the Chemical Society, Abstracts. 94 (A493): 557–558. 1908. doi:10.1039/CA9089400493.
- Semmler F (1908). "Zur Kenntnis der Bestandteile der ätherischen Öle. (Über das Elemicin, einen hochsiedenden Bestandteil des Elemiöls, und über Ersetzung von Alkyloxy-gruppen am Benzolkern durch Wasserstoff.)". Berichte der Deutschen Chemischen Gesellschaft. 41 (2): 1768–1775. doi:10.1002/cber.19080410240.
- Mauthner F (1918). "Die Synthese des Elemicins und Isoelemicins". Justus Liebigs Annalen der Chemie. 414 (2): 250–255. doi:10.1002/jlac.19184140213.
- "Synthesis of Elemicin and of isoElemicin". Journal of the Chemical Society, Abstracts. 114: i428. 1918. doi:10.1039/CA9181400421.
- Thomas L (2005). Named Organic Reactions, 2nd Edition. Wolfsburg, Germany: John Wiley & Sons, Ltd. pp. 58–60 [59]. ISBN 978-0470010402.
- Adams R (1944). "The Claisen Rearrangement" (PDF). Organic Reactions, Volume II. New York: John Wiley & Sons, Inc. pp. 2–44 [44]. doi:10.1002/0471264180.or002.01. ISBN 978-0471264187.
- Hahn G, Wassmuth H (1934). "Über β-[Oxyphenyl]-äthylamine und ihre Umwandlungen, I. Mitteil.: Synthese des Mezcalins". Berichte der Deutschen Chemischen Gesellschaft (A and B Series). 67 (4): 696–708. doi:10.1002/cber.19340670430.
- McKenna A, Nordt SP, Ryan J (August 2004). "Acute nutmeg poisoning". European Journal of Emergency Medicine. 11 (4): 240–1. doi:10.1097/01.mej.0000127649.69328.a5. PMID 15249817. S2CID 21133983.
- Shulgin AT, Sargent T, Naranjo C (December 1967). "The chemistry and psychopharmacology of nutmeg and of several related phenylisopropylamines". Psychopharmacology Bulletin. 4 (3): 13. PMID 5615546. Archived from the original (pdf) on 2014-04-20. Retrieved 2015-09-02.