Lecozotan
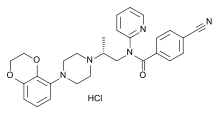
Lecozotan is an investigational drug by Wyeth tested for improvement of cognitive functions of Alzheimer's disease patients.[1] As of June 2008, the first Phase III clinical trial has been completed.[2]
 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG |
|
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C28H30ClN5O3 |
| Molar mass | 520.03 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Method of action
Lecozotan is a competitive, selective 5-HT1A receptor antagonist[3] which enhances the potassium-stimulated release of acetylcholine and glutamate.[4]
References
- Spreitzer H (August 13, 2008). "Neue Wirkstoffe - Lecozotan". Österreichische Apothekerzeitung (in German) (17/2007): 805.
- ClinicalTrials
- Schechter LE, Smith DL, Rosenzweig-Lipson S, Sukoff SJ, Dawson LA, Marquis K, et al. (September 2005). "Lecozotan (SRA-333): a selective serotonin 1A receptor antagonist that enhances the stimulated release of glutamate and acetylcholine in the hippocampus and possesses cognitive-enhancing properties". The Journal of Pharmacology and Experimental Therapeutics. 314 (3): 1274–89. arXiv:1506.05421. doi:10.1124/jpet.105.086363. PMID 15951399.
- Childers Jr WE, Harrison, BL, Abou-Gharbia, MA, Raje, S, Parks, V, Pangalos, MN, Schechter, LE (2007). "Lecozotan Hydrochloride". Drugs of the Future. 32 (5): 399–407. doi:10.1358/dof.2007.032.05.1092901.
| AChE inhibitor medications | |
|---|---|
| Other medications | |
| Experimental BACE inhibitors | |
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.