Adatanserin
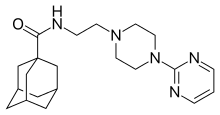
Adatanserin (WY-50,324, SEB-324) is a mixed 5-HT1A receptor partial agonist and 5-HT2A and 5-HT2C receptor antagonist.[1][2][3] It was under development by Wyeth as an antidepressant but was ultimately not pursued.[3][4]
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider |
|
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C21H31N5O |
| Molar mass | 369.513 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Adantaserin has been shown to be neuroprotective against ischemia-induced glutamatergic excitotoxicity, an effect which appears to be mediated by blockade of the 5-HT2A receptor.[5]
See also
References
- Singh A, Lucki I (April 1993). "Antidepressant-like activity of compounds with varying efficacy at 5-HT1A receptors". Neuropharmacology. 32 (4): 331–40. doi:10.1016/0028-3908(93)90153-T. PMID 8497336. S2CID 38611829.
- Kleven MS, Koek W (February 1996). "Pharmacological characterization of in vivo properties of putative mixed 5-HT1A agonist/5-HT2A/2C antagonist anxiolytics. I. Antipunishment effects in the pigeon". The Journal of Pharmacology and Experimental Therapeutics. 276 (2): 388–97. PMID 8632301.
- Abou-Gharbia MA, Childers WE, Fletcher H, et al. (December 1999). "Synthesis and SAR of adatanserin: novel adamantyl aryl- and heteroarylpiperazines with dual serotonin 5-HT(1A) and 5-HT(2) activity as potential anxiolytic and antidepressant agents". Journal of Medicinal Chemistry. 42 (25): 5077–94. doi:10.1021/jm9806704. PMID 10602693.
- Stahl, S. M. (2000). Essential psychopharmacology: neuroscientific basis and practical application. Cambridge, UK: Cambridge University Press. p. 262. ISBN 0-521-64615-4.
adatanserin.
- Dawson LA, Galandak J, Djali S (March 2002). "Attenuation of ischemic efflux of endogenous amino acids by the novel 5-HT(1A)/5-HT(2) receptor ligand adatanserin". Neurochemistry International. 40 (3): 203–9. doi:10.1016/S0197-0186(01)00082-1. PMID 11741003. S2CID 24104458.
| Simple piperazines (no additional rings) | |
|---|---|
| Phenylpiperazines |
|
| Benzylpiperazines | |
| Diphenylalkylpiperazines (benzhydrylalkylpiperazines) |
|
| Pyrimidinylpiperazines | |
| Pyridinylpiperazines | |
| Benzo(iso)thiazolylpiperazines | |
| Tricyclics (piperazine attached via side chain) |
|
| Others/Uncategorized | |
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.