Smoothened
Smoothened is a protein that in humans is encoded by the SMO gene. Smoothened is a Class Frizzled (Class F) G protein-coupled receptor[5][6] that is a component of the hedgehog signaling pathway and is conserved from flies to humans. It is the molecular target of the natural teratogen cyclopamine.[7] It also is the target of Vismodegib, the first hedgehog pathway inhibitor to be approved by the U.S. Food and Drug Administration (FDA).[8]
Smoothened (Smo) is a key transmembrane protein that is a key component of the hedgehog signaling pathway, a cell-cell communication system critical for embryonic development and adult tissue homeostasis.[9][10] Mutations in proteins that relay Hh signals between cells cause birth defects and cancer.[11] The protein that carries the Hh signal across the membrane is the oncoprotein and G-protein coupled receptor (GPCR) Smoothened (Smo). Smo is regulated by a separate transmembrane receptor for Hh ligands called Patched (Ptc). Ptc itself is a tumor suppressor that keeps the Hh pathway off by inhibiting Smo. The excessive Hh signaling that drives human skin and brain cancer is most frequently caused by inactivating mutations in Ptc or by gain of function mutations in Smo. While direct Smo agonists and antagonists, such as SAG and vismodegib, can bind to and activate or inhibit Smo, how Ptc inhibits Smo endogenously remains a mystery in the field.
Currently, Smo is targeted and inhibited directly by a small-molecule drug, vismodegib, for the treatment of advanced basal cell cancer, however widespread resistance to this drug has become a prevalent issue.[12][13] Finding another method to target Smo activity in Hh-driven cancers would provide valuable information for novel therapeutics. Identifying these Ptc responsive sites on Smo will help solve a long-standing mystery in Hh signaling and suggest new therapeutic strategies to block Smo activity in Hh-driven cancers.
Function
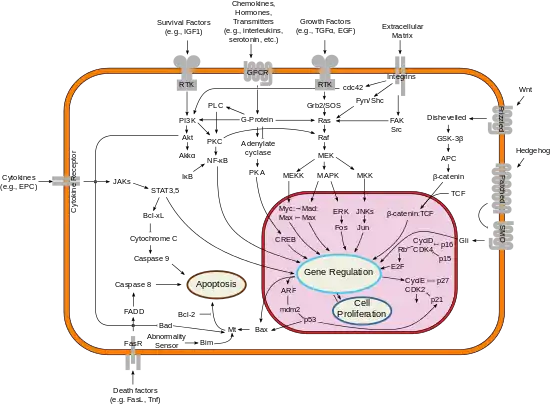
Cellular localization plays an essential role in the function of SMO, which anchors to the cell membrane as a 7-pass transmembrane protein. Stimulation of the patched 12-pass transmembrane receptor by the sonic hedgehog ligand leads to translocation of SMO to the primary cilium in vertebrates in a process that involves the exit of patched from the primary cilium, where it normally localizes in its unstimulated state.[14] Vertebrate SMO that is mutated in the domain required for ciliary localisation often cannot contribute to hedgehog pathway activation.[15] Conversely, SMO can become constitutively localized to the primary cilium and potentially activate pathway signaling constitutively as a result of a tryptophan to leucine mutation in the aforementioned domain.[16] SMO has been shown to move during patched stimulation from the plasma membrane near the primary cilium to the ciliary membrane itself via a lateral transport pathway along the membrane, as opposed to via directed transport by vesicles. The cAMP-PKA pathway is known to promote the lateral movement of SMO and hedgehog signal transduction in general.[17] In invertebrates like Drosophila, SMO does not organize at cilia and instead is generally translocated to the plasma membrane following hedgehog binding to patched.[18]
After cellular localization, SMO must additionally be activated by a distinct mechanism in order to stimulate hedgehog signal transduction, but that mechanism is unknown.[19] There is evidence for the existence of an unidentified endogenous ligand that binds SMO and activates it. It is believed that mutations in SMO can mimic the ligand-induced conformation of SMO and activate constitutive signal transduction.[18]
SMO plays a key role in transcriptional repression and activation by the zinc-finger transcription factor Cubitus interruptus (Ci; known as Gli in vertebrates). When the hedgehog pathway is inactive, a complex of Fused (Fu), Suppressor of Fused (Sufu), and the kinesin motor protein Costal-2 (Cos2) tether Ci to microtubules. In this complex, Cos2 promotes proteolytic cleavage of Ci by activating hyperphosphorylation of Ci and subsequent recruitment of ubiquitin ligase; the cleaved Ci goes on to act as a repressor of hedgehog-activated transcription. However, when hedgehog signaling is active, Ci remains intact and acts as a transcriptional activator of the same genes that its cleaved form suppresses.[20][21] SMO has been shown to bind Costal-2 and play a role in the localization of the Ci complex and prevention of Ci cleavage.[22][23] Additionally, it is known that vertebrate SMO contributes to the activation of Gli as a transcription factor via association with ciliary structures such as Evc2, but these mechanisms are not fully understood.[18]
Endogenous Activation
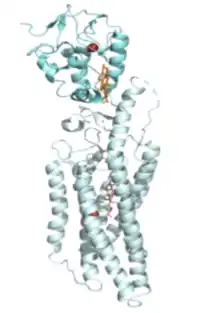
A leading hypothesis in the field is that Ptc regulates Smo by gating its access to cholesterol or a related sterol.[24] It has been proposed that cholesterol activates Smo, and subsequently Hh signaling, by entering the active site through a hydrophobic “oxysterol tunnel,” which can adopt open or closed conformations to allow for activation or inactivation of Smo, respectively, due to allowed sterol binding.[25][26] Shh would work by inhibiting Ptc, which would increase accessible cholesterol concentrations and allow for the activation of Smo anf transmission of the Hh signal.[27] A recent crystal structure has identified two sterol binding sites in Smo, but which site is endogenously regulated by Ptc remains to be determined. The potential sites of regulation include the extracellular cysteine-rich domain (CRD) of Smo, as well as a site deep within the transmembrane domain (TMD).[28][29][30]
Due to the abundance of cholesterol in the plasma membrane (up to 50 mole %), it has also been proposed that Ptc regulates the activity of Smo by controlling cholesterol accessibility specifically within the membrane of the primary cilia, which contains a less abundant, and therefore more readily regulated pool of accessible cholesterol.[28][31]
Typically, upon activation and release of inhibition by Ptc, Smo will relocate to the primary cilia and Ptc will diffuse out of the ciliary membrane.[32] Upon inactivation, Smo no longer becomes concentrated in the ciliary membrane, This hypothesis is supported by methods which can increase or deplete the accessible cholesterol pool, with a subsequent increase or decrease in Hh signaling. This accessible cholesterol pool has been shown to be distinct from the general plasma membrane cholesterol pool in being available for protein interaction and cell uptake. The ciliary membrane has also been shown to contain lower levels of accessible cholesterol due to sequestering of cholesterol by sphingomyelin. In addition to cholesterol’s role as a Hh pathway agonist, it has been shown that cholesterol levels within the ciliary membrane rapidly increase upon treatment with Shh only in the presence of Ptc, further suggesting Ptc regulation of accessible cholesterol as the mechanism behind Smo activation/inhibition.[27] Additionally, Molecular Dynamics simulations suggest that vismodegib inhibits Smo through a conformational change that prevents cholesterol from binding.[33] This suggests the hypothesis that Ptc functions by preventing Smo access to cholesterol, and upon Ptc inhibition by Shh, Smo gains access to cholesterol and is subsequently activated, transmitting the Hh signal.
Role in disease
SMO can function as an oncogene. Activating SMO mutations can lead to unregulated activation of the hedgehog pathway and serve as driving mutations for cancers such as medulloblastoma, basal-cell carcinoma, pancreatic cancer, and prostate cancer.[16][34] As such, SMO is an attractive cancer drug target, along with the many hedgehog pathway agonists and antagonists that are known to directly target SMO.[16]
Cholesterol is known to be crucial in regulating the overall hedgehog pathway, and congenital mutations in cholesterol synthesis pathways can inactivate SMO specifically, leading to developmental disorders.[35] For example, oxysterol 20(S)-OHC is known to activate vertebrate SMO by binding the cysteine rich domain near its extracellular amino-terminal region. In the context of cancer, 20(S)-OHC is the target of a proposed anti-cancer oxysterol binding inhibitor.[18]
Agonists
- Smoothened agonist (SAG)
- Purmorphamine
- Oxysterols including 20(S)-OHC and 20(S)-yne
Antagonists
- Cyclopamine
- Itraconazole
- Vismodegib (Erivedge), a smoothened receptor inhibitor for the treatment of basal-cell carcinoma, being investigated for the treatment of other types of cancer
- SANT-1
- Sonidegib
- Patidegib[36]
- Glasdegib
- Jervine
See also
References
- GRCh38: Ensembl release 89: ENSG00000128602 - Ensembl, May 2017
- GRCm38: Ensembl release 89: ENSMUSG00000001761 - Ensembl, May 2017
- "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- Ruiz-Gómez A, Molnar C, Holguín H, Mayor F, de Celis JF (April 2007). "The cell biology of Smo signalling and its relationships with GPCRs". Biochimica et Biophysica Acta (BBA) - Biomembranes. 1768 (4): 901–12. doi:10.1016/j.bbamem.2006.09.020. PMID 17094938.
- Wang C, Wu H, Evron T, Vardy E, Han GW, Huang XP, Hufeisen SJ, Mangano TJ, Urban DJ, Katritch V, Cherezov V, Caron MG, Roth BL, Stevens RC (July 2014). "Structural basis for Smoothened receptor modulation and chemoresistance to anticancer drugs". Nature Communications. 5: 4355. Bibcode:2014NatCo...5.4355W. doi:10.1038/ncomms5355. PMC 4198951. PMID 25008467.
- Taipale J, Chen JK, Cooper MK, Wang B, Mann RK, Milenkovic L, Scott MP, Beachy PA (August 2000). "Effects of oncogenic mutations in Smoothened and Patched can be reversed by cyclopamine". Nature. 406 (6799): 1005–9. Bibcode:2000Natur.406.1005T. doi:10.1038/35023008. PMID 10984056. S2CID 4313790.
- "Vismodegib, First Hedgehog Inhibitor, Approved for BCC Patients". OncLive. Retrieved 2017-05-26.
- Kong JH, Siebold C, Rohatgi R (May 2019). "Biochemical mechanisms of vertebrate hedgehog signaling". Development. 146 (10): dev166892. doi:10.1242/dev.166892. PMC 6550017. PMID 31092502.
- Jeng KS, Chang CF, Lin SS (January 2020). "Sonic Hedgehog Signaling in Organogenesis, Tumors, and Tumor Microenvironments". Int J Mol Sci. 21 (3): 758. doi:10.3390/ijms21030758. PMC 7037908. PMID 31979397.
- Liu X, Ding C, Tan W, Zhang A (February 2020). "Medulloblastoma: Molecular understanding, treatment evolution, and new developments". Pharmacol. Ther. 210: 107516. doi:10.1016/j.pharmthera.2020.107516. PMID 32105673.
- Laliscia C, Baldaccini D, Antonuzzo A, Paiar F (2019). "Vismodegibfor the treatment of radiation-induced basal cell carcinoma - a case report and brief literature study". Contemp Oncol (Pozn). 23 (4): 251–253. doi:10.5114/wo.2019.91540. PMC 6978755. PMID 31992959.
- Liao S, Floyd C, Verratti N, Leung L, Wu C (March 2020). "Analysis of vismodegib resistance in D473G and W535L mutants of SMO receptor and design of novel drug derivatives using molecular dynamics simulations". Life Sci. 244: 117302. doi:10.1016/j.lfs.2020.117302. PMID 31953165.
- Rohatgi R, Milenkovic L, Scott MP (July 2007). "Patched1 regulates hedgehog signaling at the primary cilium". Science. 317 (5836): 372–6. Bibcode:2007Sci...317..372R. doi:10.1126/science.1139740. PMID 17641202.
- Corbit KC, Aanstad P, Singla V, Norman AR, Stainier DY, Reiter JF (October 2005). "Vertebrate Smoothened functions at the primary cilium". Nature. 437 (7061): 1018–21. Bibcode:2005Natur.437.1018C. doi:10.1038/nature04117. PMID 16136078. S2CID 4431804.
- Wang Y, Zhou Z, Walsh CT, McMahon AP (February 2009). "Selective translocation of intracellular Smoothened to the primary cilium in response to Hedgehog pathway modulation". Proceedings of the National Academy of Sciences of the United States of America. 106 (8): 2623–8. Bibcode:2009PNAS..106.2623W. doi:10.1073/pnas.0812110106. PMC 2650314. PMID 19196978.
- Milenkovic L, Scott MP, Rohatgi R (November 2009). "Lateral transport of Smoothened from the plasma membrane to the membrane of the cilium". The Journal of Cell Biology. 187 (3): 365–74. doi:10.1083/jcb.200907126. PMC 2779247. PMID 19948480.
- Arensdorf AM, Marada S, Ogden SK (January 2016). "Smoothened Regulation: A Tale of Two Signals". Trends in Pharmacological Sciences. 37 (1): 62–72. doi:10.1016/j.tips.2015.09.001. PMC 4593303. PMID 26432668.
- Rohatgi R, Milenkovic L, Corcoran RB, Scott MP (March 2009). "Hedgehog signal transduction by Smoothened: pharmacologic evidence for a 2-step activation process". Proceedings of the National Academy of Sciences of the United States of America. 106 (9): 3196–201. Bibcode:2009PNAS..106.3196R. doi:10.1073/pnas.0813373106. PMC 2642660. PMID 19218434.
- Cohen MM (November 2003). "The hedgehog signaling network". American Journal of Medical Genetics. Part A. 123A (1): 5–28. doi:10.1002/ajmg.a.20495. PMID 14556242. S2CID 31906029.
- Aikin RA, Ayers KL, Thérond PP (April 2008). "The role of kinases in the Hedgehog signalling pathway". EMBO Reports. 9 (4): 330–6. doi:10.1038/embor.2008.38. PMC 2288774. PMID 18379584.
- Lum L, Zhang C, Oh S, Mann RK, von Kessler DP, Taipale J, Weis-Garcia F, Gong R, Wang B, Beachy PA (November 2003). "Hedgehog signal transduction via Smoothened association with a cytoplasmic complex scaffolded by the atypical kinesin, Costal-2". Molecular Cell. 12 (5): 1261–74. doi:10.1016/S1097-2765(03)00426-X. PMID 14636583.
- Jia J, Tong C, Jiang J (November 2003). "Smoothened transduces Hedgehog signal by physically interacting with Costal2/Fused complex through its C-terminal tail". Genes & Development. 17 (21): 2709–20. doi:10.1101/gad.1136603. PMC 280620. PMID 14597665.
- Hu A, Song BL (December 2019). "The interplay of Patched, Smoothened and cholesterol in Hedgehog signaling". Curr. Opin. Cell Biol. 61: 31–38. doi:10.1016/j.ceb.2019.06.008. PMID 31369952.
- Deshpande I, Liang J, Hedeen D, Roberts KJ, Zhang Y, Ha B, Latorraca NR, Faust B, Dror RO, Beachy PA, Myers BR, Manglik A (July 2019). "Smoothened stimulation by membrane sterols drives Hedgehog pathway activity". Nature. 571 (7764): 284–288. doi:10.1038/s41586-019-1355-4. PMC 6709672. PMID 31263273.
- Kowatsch C, Woolley RE, Kinnebrew M, Rohatgi R, Siebold C (August 2019). "Structures of vertebrate Patched and Smoothened reveal intimate links between cholesterol and Hedgehog signalling". Curr. Opin. Struct. Biol. 57: 204–214. doi:10.1016/j.sbi.2019.05.015. PMC 6744280. PMID 31247512.
- Kinnebrew M, Iverson EJ, Patel BB, Pusapati GV, Kong JH, Johnson KA, Luchetti G, Eckert KM, McDonald JG, Covey DF, Siebold C, Radhakrishnan A, Rohatgi R (October 2019). "Cholesterol accessibility at the ciliary membrane controls hedgehog signaling". eLife. 8. doi:10.7554/eLife.50051. PMC 6850779. PMID 31657721.
- Luchetti G, Sircar R, Kong JH, Nachtergaele S, Sagner A, Byrne EF, Covey DF, Siebold C, Rohatgi R (October 2016). "Cholesterol activates the G-protein coupled receptor Smoothened to promote Hedgehog signaling". eLife. 5. doi:10.7554/eLife.20304. PMC 5123864. PMID 27705744.
-
- Hedger G, Koldsø H, Chavent M, Siebold C, Rohatgi R, Sansom MSP (March 2019). "Cholesterol Interaction Sites on the Transmembrane Domain of the Hedgehog Signal Transducer and Class F G Protein-Coupled Receptor Smoothened". Structure. 27 (3): 549–559.e2. doi:10.1016/j.str.2018.11.003. PMC 6408332. PMID 30595453.
- Byrne EF, Luchetti G, Rohatgi R, Siebold C (April 2018). "Multiple ligand binding sites regulate the Hedgehog signal transducer Smoothened in vertebrates". Curr. Opin. Cell Biol. 51: 81–88. doi:10.1016/j.ceb.2017.10.004. PMC 5949240. PMID 29268141.
- Gigante ED, Caspary T (February 2020). "Signaling in the primary cilium through the lens of the Hedgehog pathway". Wiley Interdiscip Rev Dev Biol. 9 (6): e377. doi:10.1002/wdev.377. PMC 7444278. PMID 32084300.
-
- Weiss LE, Milenkovic L, Yoon J, Stearns T, Moerner WE (March 2019). "Motional dynamics of single Patched1 molecules in cilia are controlled by Hedgehog and cholesterol". Proc. Natl. Acad. Sci. U.S.A. 116 (12): 5550–5557. doi:10.1073/pnas.1816747116. PMC 6431229. PMID 30819883.
- An X, Bai Q, Bai F, Shi D, Liu H, Yao X (2019). "Deciphering the Allosteric Effect of Antagonist Vismodegib on Smoothened Receptor Deactivation Using Metadynamics Simulation". Front Chem. 7: 406. Bibcode:2019FrCh....7..406Y. doi:10.3389/fchem.2019.00406. PMC 6558189. PMID 31214579.
- Xie J, Murone M, Luoh SM, Ryan A, Gu Q, Zhang C, Bonifas JM, Lam CW, Hynes M, Goddard A, Rosenthal A, Epstein EH, de Sauvage FJ (January 1998). "Activating Smoothened mutations in sporadic basal-cell carcinoma". Nature. 391 (6662): 90–2. Bibcode:1998Natur.391...90X. doi:10.1038/34201. PMID 9422511. S2CID 205003240.
- Blassberg R, Macrae JI, Briscoe J, Jacob J (February 2016). "Reduced cholesterol levels impair Smoothened activation in Smith-Lemli-Opitz syndrome". Human Molecular Genetics. 25 (4): 693–705. doi:10.1093/hmg/ddv507. PMC 4743690. PMID 26685159.
- Patidegib
Further reading
- Chen Y, Struhl G (November 1996). "Dual roles for patched in sequestering and transducing Hedgehog". Cell. 87 (3): 553–63. doi:10.1016/S0092-8674(00)81374-4. PMID 8898207. S2CID 15208834.
- Stone DM, Hynes M, Armanini M, Swanson TA, Gu Q, Johnson RL, Scott MP, Pennica D, Goddard A, Phillips H, Noll M, Hooper JE, de Sauvage F, Rosenthal A (November 1996). "The tumour-suppressor gene patched encodes a candidate receptor for Sonic hedgehog". Nature. 384 (6605): 129–34. Bibcode:1996Natur.384..129S. doi:10.1038/384129a0. PMID 8906787. S2CID 4342540.
- Xie J, Murone M, Luoh SM, Ryan A, Gu Q, Zhang C, Bonifas JM, Lam CW, Hynes M, Goddard A, Rosenthal A, Epstein EH, de Sauvage FJ (January 1998). "Activating Smoothened mutations in sporadic basal-cell carcinoma". Nature. 391 (6662): 90–2. Bibcode:1998Natur.391...90X. doi:10.1038/34201. PMID 9422511. S2CID 205003240.
- Reifenberger J, Wolter M, Weber RG, Megahed M, Ruzicka T, Lichter P, Reifenberger G (May 1998). "Missense mutations in SMOH in sporadic basal cell carcinomas of the skin and primitive neuroectodermal tumors of the central nervous system". Cancer Research. 58 (9): 1798–803. PMID 9581815.
- Sublett JE, Entrekin RE, Look AT, Reardon DA (May 1998). "Chromosomal localization of the human smoothened gene (SMOH) to 7q32. 3 by fluorescence in situ hybridization and radiation hybrid mapping". Genomics. 50 (1): 112–4. doi:10.1006/geno.1998.5227. PMID 9628830.
- McGarvey TW, Maruta Y, Tomaszewski JE, Linnenbach AJ, Malkowicz SB (September 1998). "PTCH gene mutations in invasive transitional cell carcinoma of the bladder". Oncogene. 17 (9): 1167–72. doi:10.1038/sj.onc.1202045. PMID 9764827.
- Chidambaram A, Gerrard B, Hanson M, Dean M (November 1998). "Chromosomal localization of the human and murine orthologues of the Drosophila smoothened gene". Genomics. 53 (3): 416–7. doi:10.1006/geno.1998.5531. PMID 9799615.
- Carpenter D, Stone DM, Brush J, Ryan A, Armanini M, Frantz G, Rosenthal A, de Sauvage FJ (November 1998). "Characterization of two patched receptors for the vertebrate hedgehog protein family". Proceedings of the National Academy of Sciences of the United States of America. 95 (23): 13630–4. Bibcode:1998PNAS...9513630C. doi:10.1073/pnas.95.23.13630. PMC 24870. PMID 9811851.
- Detmer K, Walker AN, Jenkins TM, Steele TA, Dannawi H (August 2000). "Erythroid differentiation in vitro is blocked by cyclopamine, an inhibitor of hedgehog signaling". Blood Cells, Molecules & Diseases. 26 (4): 360–72. doi:10.1006/bcmd.2000.0318. PMID 11042037.
- Long F, Zhang XM, Karp S, Yang Y, McMahon AP (December 2001). "Genetic manipulation of hedgehog signaling in the endochondral skeleton reveals a direct role in the regulation of chondrocyte proliferation". Development. 128 (24): 5099–108. PMID 11748145.
- Incardona JP, Gruenberg J, Roelink H (June 2002). "Sonic hedgehog induces the segregation of patched and smoothened in endosomes". Current Biology. 12 (12): 983–95. doi:10.1016/S0960-9822(02)00895-3. PMID 12123571. S2CID 3162414.
- Lowrey JA, Stewart GA, Lindey S, Hoyne GF, Dallman MJ, Howie SE, Lamb JR (August 2002). "Sonic hedgehog promotes cell cycle progression in activated peripheral CD4(+) T lymphocytes". Journal of Immunology. 169 (4): 1869–75. doi:10.4049/jimmunol.169.4.1869. PMID 12165511.
- Taipale J, Cooper MK, Maiti T, Beachy PA (August 2002). "Patched acts catalytically to suppress the activity of Smoothened". Nature. 418 (6900): 892–7. Bibcode:2002Natur.418..892T. doi:10.1038/nature00989. PMID 12192414. S2CID 4362029.
- Katayam M, Yoshida K, Ishimori H, Katayama M, Kawase T, Motoyama J, Kamiguchi H (September 2002). "Patched and smoothened mRNA expression in human astrocytic tumors inversely correlates with histological malignancy". Journal of Neuro-Oncology. 59 (2): 107–15. doi:10.1023/A:1019660421216. PMID 12241103. S2CID 21237084.
- Couvé-Privat S, Bouadjar B, Avril MF, Sarasin A, Daya-Grosjean L (December 2002). "Significantly high levels of ultraviolet-specific mutations in the smoothened gene in basal cell carcinomas from DNA repair-deficient xeroderma pigmentosum patients". Cancer Research. 62 (24): 7186–9. PMID 12499255.
- Grachtchouk V, Grachtchouk M, Lowe L, Johnson T, Wei L, Wang A, de Sauvage F, Dlugosz AA (June 2003). "The magnitude of hedgehog signaling activity defines skin tumor phenotype". The EMBO Journal. 22 (11): 2741–51. doi:10.1093/emboj/cdg271. PMC 156767. PMID 12773389.
- Chen W, Ren XR, Nelson CD, Barak LS, Chen JK, Beachy PA, de Sauvage F, Lefkowitz RJ (December 2004). "Activity-dependent internalization of smoothened mediated by beta-arrestin 2 and GRK2". Science. 306 (5705): 2257–60. Bibcode:2004Sci...306.2257C. doi:10.1126/science.1104135. PMID 15618519. S2CID 12823611.
- Byrne EF, Sircar R, Miller PS, Hedger G, Luchetti G, Nachtergaele S, Tully MD, Mydock-McGrane L, Covey DF, Rambo RP, Sansom MS, Newstead S, Rohatgi R (July 2016). "Structural basis of Smoothened regulation by its extracellular domains". Nature. 535 (7613): 517–22. Bibcode:2016Natur.535..517B. doi:10.1038/nature18934. PMC 4970916. PMID 27437577.
External links
- "Frizzled Receptors: SMO". IUPHAR Database of Receptors and Ion Channels. International Union of Basic and Clinical Pharmacology.
- SMO+protein,+human at the US National Library of Medicine Medical Subject Headings (MeSH)